Key Highlights
- Due to a shorter life expectancy than other oncology patients, neuro-oncological patients' treatment cannot be much delayed, regardless of the COVID-19 infection. According to an article published by Acta bio-medica: Atenei Parmensis in July 2022, the international scientific community urgently reorganized the treatment approach to minimize the risk of in-hospital contagious. For glioblastoma multiforme patients, adjuvant treatments were evaluated, and a hypofractionated radiotherapy regimen was preferred to reduce daily hospital access. Furthermore, the above source mentioned that the patient-doctor relationship improved through telemedicine. Thus, telemedicine benefits in terms of financial consequences for the health system, psychological satisfaction of the patient, and equate medical care for glioblastoma multiforme patients and led to an increase in glioblastoma multiforme treatment, thereby driving the market growth.
- Factors such as the increasing burden of brain disorders, strong R&D initiatives from key players, and a rising geriatric population are boosting the growth of the glioblastoma multiforme treatment market. For instance, according to an article published by the National Brain Tumor Society, more than 13,000 Americans are expected to receive a glioblastoma multiforme diagnosis in 2022. Glioblastoma multiforme accounts for 49.1% of all primary malignant brain tumors. It is estimated that more than 10,000 individuals in the United States will succumb to glioblastoma every year. Thus, with increased glioblastoma multiforme cases, the demand for its treatment increases, leading to market growth.
- Moreover, various initiatives undertaken by the regional and state governments to make the treatment more accessible to patients are anticipated to boost the market growth further. For instance, in December 2022, the Food and Drug Administration (FDA) granted Fast Track designation to WP1122 for the treatment of glioblastoma multiforme. WP1122 was a prodrug of 2-deoxy-D-glucose, or 2-DG, designed to target glycolysis and glycosylation, limiting viral infectivity and replication. Such an initiative is expected to increase the demand for glioblastoma multiforme treatment, thereby driving market growth.
- Hence, as per the abovementioned factors, the glioblastoma multiforme treatment market is anticipated to grow over the forecast period. However, reimbursement issues, stringent regulatory guidelines, and high cost & side effects associated with therapies are expected to restrain the growth of the global market over the analysis period.
Glioblastoma Multiforme Treatment Market Trends
Chemotherapy Segment is Expected to Dominate the Market Over the Forecast Period
- The key factors contributing to the growth of the radiation therapy segment are the rising number of brain and other nervous system cancers and the growing preference and commercial availability of chemotherapeutic agents for treating glioblastoma. For instance, according to an article published by thThe key factors contributing to the growth of the radiation therapy segment are the rising number of brain and other nervous system cancers and the growing preference and commercial availability of chemotherapeutic agents for treating glioblastoma. For instance, according to an article published by the Chinese Clinical Oncology in December 2022, radiotherapy continues to be an important component of the treatment of glioma, particularly high-grade glioma and glioblastoma multiforme. Radiotherapy techniques have improved with time, and many options have become available in the interest of minimizing adverse effects and improving survival rates and quality of life. Proton therapy (PT), or proton beam radiation therapy (PBRT), offers certain advantages to other modern photon-based conformal therapy when used in the treatment of CNS malignancies such as glioblastoma multiforme. Thus, the adoption of radiotherapy due to its advantage is expected to drive segmental growth.
- Furthermore, increasing developments and launches by manufacturers to meet the growing demand for innovative products are expected to drive market growth. For instance, in January 2022, ITM Isotope Technologies Munich SE reported a cooperation agreement for the clinical development of a radiopharmaceutical therapy candidate to treat malignant brain tumor glioblastoma. ITM and Helmholtz Munich collaborated to support an upcoming dose-escalation Phase I clinical trial with LuCaFab (ITM-31). LuCaFab is a CA XII-specific antibody Fab fragment targeting molecule, developed by Helmholtz Munich, radiolabeled with ITM's medical radioisotope no-carrier-added lutetium-177 (n.c.a. 177Lu, EndolucinBeta).
- Thus, all the aforementioned factors are anticipated to boost the segment over the forecast period.e Chinese Clinical Oncology in December 2022, radiotherapy continues to be an important component of the treatment of glioma, particularly high-grade glioma, and glioblastoma multiforme. Radiotherapy techniques have improved with time, and many options have become available in the interest of minimizing adverse effects and improving survival rates and quality of life. Proton therapy (PT), or proton beam radiation therapy (PBRT), offers certain advantages to other modern photon-based conformal therapy when used in the treatment of CNS malignancies such as glioblastoma multiforme. Thus, the adoption of radiotherapy due to its advantage is expected to drive segmental growth.
- Furthermore, increasing developments and launches by manufacturers to meet the growing demand for innovative products are expected to drive market growth. For instance, in January 2022, ITM Isotope Technologies Munich SE reported a cooperation agreement for the clinical development of a radiopharmaceutical therapy candidate to treat malignant brain tumor glioblastoma. ITM and Helmholtz Munich collaborated to support an upcoming dose-escalation Phase I clinical trial with LuCaFab (ITM-31). LuCaFab is a CA XII-specific antibody Fab fragment targeting molecule, developed by Helmholtz Munich, radiolabeled with ITM's medical radioisotope no-carrier-added lutetium-177 (n.c.a. 177Lu, EndolucinBeta).
- Thus, all the aforementioned factors are anticipated to boost the segment over the forecast period.
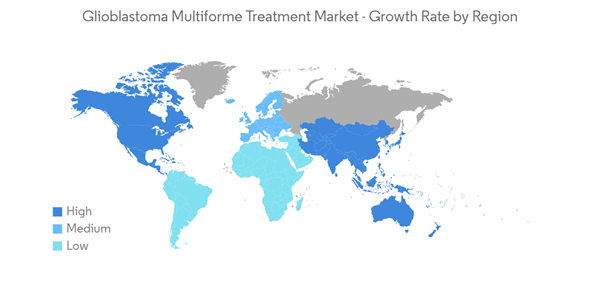
North America leads the Glioblastoma Multiforme Treatment Market Over the Forecast Period
- North America leads the glioblastoma multiforme treatment market, owing to a rise in funding initiatives and awareness among the patient population. Government support for the development of the healthcare sector, widespread awareness of brain disorders and tumors, easy access to high-quality medical facilities, and favorable reimbursement policies are among the key factors driving the regional market's growth.
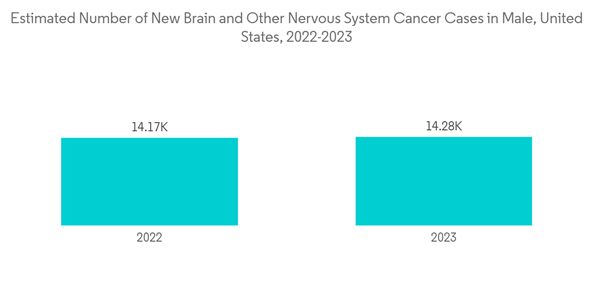
- According to the American Society of Clinical Oncology (ASCO), in February 2022, it was expected that an estimated 25,050 adults (14,170 men and 10,880 women) in the United States would be diagnosed with primary cancerous tumors of the brain and spinal cord in 2022. Additionally, as per the above source, brain tumors account for 85% to 90% of all primary CNS tumors. Moreover, about 4,170 children under the age of 15 were diagnosed with a brain or CNS tumor this year in the United States.
- Furthermore, strategic activities by the key market players in the region are anticipated to boost the market's growth. For instance, in July 2022, Diffusion Pharmaceuticals Inc. reported that after collaboration with the United States Food and Drug Administration on the design of their Phase 2 clinical trial entitled 'Open-Label, Dose-Escalation, Phase 2 Safety and Efficacy Study of TSC in Newly Diagnosed Glioblastoma ('GBM') Patients when Administered with Standard of Care.
- Thus, given the aforementioned factors, the glioblastoma multiforme treatment market is expected to grow significantly over the forecast period in North America.
Glioblastoma Multiforme Treatment Industry Overview
The glioblastoma multiforme treatment market is moderately concentrated in nature due to the presence of companies operating globally as well as regionally. The competitive landscape includes an analysis of a few international as well as local companies that hold market shares and are well known, including Arbor Pharmaceuticals, Bristol-Myers Squibb, Eli Lilly and Company, F. Hoffmann-La Roche Ltd, Merck & Co. Inc., Sun Pharmaceuticals, and Teva Pharmaceutical Industries Ltd.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Arbor Pharmaceuticals, LLC
- Bristol-Myers Squibb Company
- Eli Lilly and Company
- F. Hoffmann-La Roche Ltd
- Merck & Co. Inc.
- Sun Pharmaceutical Industries Ltd.
- Lanthen Pharmaceutical
- Pfizer Inc.
- Amgen Inc.
- Teva Pharmaceutical Industries Ltd.