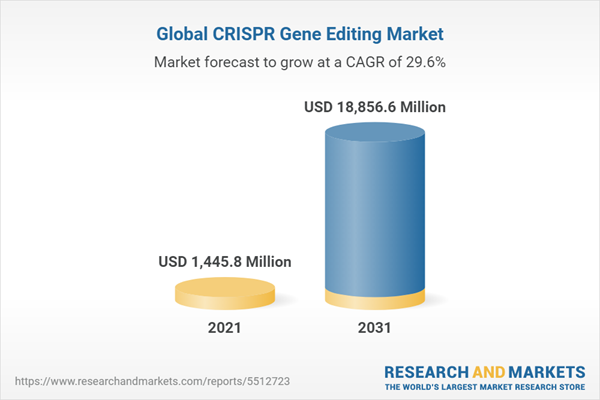
The global CRISPR gene editing market was valued at $1,088.6 million in 2020, and it is expected to reach $18,856.6 million by 2031, registering a CAGR of 29.60% during the forecast period.
Market Report Coverage - CRISPR Gene Editing
Market Segmentation
- Product Type - CRISPR Products (Kits and Enzymes, Libraries, Design Tools, Antibodies, and Other Products), CRISPR Services (gRNA Design & Vector Construction, Cell Line and Engineering, Screening Services, and Other Services)
- Application - Agriculture, Biomedical (Gene Therapy, Drug Discovery, And Diagnostics), Industrial, and Other Applications
- End User - Academic Institutes and Research Centers, Biotechnology Companies, Contract Research Organizations (CROs), and Pharmaceutical and Biopharmaceutical Companies
Regional Segmentation
- North America - U.S., Canada
- Europe - Germany, France, Italy, U.K., Spain, Switzerland, and Rest-of-Europe
- Asia-Pacific - China, Japan, India, South Korea, Singapore, Australia, and Rest-of-Asia-Pacific (RoAPAC)
- Latin America - Brazil, Mexico, and Rest-of-Latin America
- Rest-of-the-World
Market Growth Drivers
- Prevalence of Genetic Disorders and Use of Gene Editing
- Government and Private Funding
- Technology Advancement in CRISPR Gene Editing
Market Challenges
- Off-Target Effects and Delivery
- Ethical Concerns and Implications with Respect to Human Gene Editing
Market Opportunities
- Expanding Gene and Cell Therapy Area
- CRISPR Gene Editing Scope in Agriculture
Key Companies Profiled
Abcam, Inc., Applied StemCell, Inc., Agilent Technologies, Inc., Cellecta, Inc., CRISPR Therapeutics AG, Thermo Fisher Scientific, Inc., GeneCopoeia, Inc., GenScript Biotech Corporation, Horizon Discovery Group PLC, Integrated DNA Technologies, Inc., Merck KGaA, New England Biolabs, Inc., Origene Technologies, Inc., Rockland Immunochemicals, Inc., Synthego Corporation, System Biosciences LLC, ToolGen, Inc., Takara Bio
Key Questions Answered in this Report:
- What is CRISPR gene editing?
- What is the timeline for the development of CRISPR technology?
- How did the CRISPR gene editing market evolve, and what is its scope in the future?
- How is the CRISPR gene editing clinical trial pipeline advancing?
- What are the major market drivers, restraints, and opportunities in the global CRISPR gene editing market?
- What are the key developmental strategies that are being implemented by key players to sustain this market?
- What is the patent landscape of this market? What will be the impact of patent expiry on this market?
- What is the impact of COVID-19 on this market?
- What are the guidelines implemented by different government bodies to regulate the approval of CRISPR products/therapies?
- How is CRISPR gene editing being utilized for the development of therapeutics?
- How will the investments by public and private companies and government organizations affect the global CRISPR gene editing market?
- What was the market size of the leading segments and sub-segments of the global CRISPR gene editing market in 2020?
- How will the industry evolve during the forecast period 2021-2031?
- What will be the growth rate of the CRISPR gene editing market during the forecast period 2021-2031?
- How will each of the segments of the global CRISPR gene editing market grow during the forecast period, and what will be the revenue generated by each of the segments by the end of 2031?
- Which product and application segments are expected to register the highest CAGR for the global CRISPR gene editing market?
- What are the major benefits of the implementation of CRISPR gene editing in different fields of applications, including biomedical research, agricultural research, industrial research, gene therapy, drug discovery, and diagnostics?
- What is the market size of the CRISPR gene editing market in different countries of the world?
- Which geographical region is expected to contribute to the highest sales of the CRISPR gene editing market?
- What are the reimbursement scenario and regulatory structure for the CRISPR gene editing market in different regions?
- What are the key strategies incorporated by the players of the global CRISPR gene editing market to sustain the competition and retain their supremacy?
Market Overview
The development of genome engineering with potential applications proved to reflect a remarkable impact on the future of the healthcare and life science industry. The high efficiency of the CRISPR-Cas9 system has been demonstrated in various studies for genome editing, which resulted in significant investments within the field of genome engineering. However, with so many advantages, limitations also exist, which need consideration before clinical applications. Further, many researchers are working on the limitations of CRISPR gene editing technology for better results. The potential of CRISPR gene editing to alter the human genome and modify the disease conditions is incredible but exists with ethical and social concerns. The global CRISPR gene editing market was valued at $1,088.6 million in 2020, and it is expected to reach $18,856.6 million by 2031, registering a CAGR of 29.60% during the forecast period.
The growth is attributed to the increasing demand in the food industry for better products with improved quality and nutrient enrichment and the pharmaceutical industry for targeted treatment for various diseases. Further, the continued significant investments by healthcare companies to meet the industry demand and growing prominence for the gene therapy procedures with less turnaround time are the prominent factors propelling the growth of the global CRISPR gene editing market.
Research organizations, pharmaceutical and biotechnology industries, and institutes are looking for more efficient genome editing technologies to increase specificity and cost-effectiveness, and reduce turnaround time and human errors. Further, the evolution of genome editing technologies has enabled the exploration of a wide range of applications in various fields, such as industrial, biotechnology, and agricultural research. These advanced methods are simple, super-efficient, cost-effective, provide multiplexing and high throughput capabilities. The rising geriatric population and the increasing number of cancer cases and genetic disorders across the globe are expected to translate into significantly higher demand for the CRISPR gene editing market.
Furthermore, the companies are investing a huge amount in researching and developing CRISPR gene editing products and gene therapies. The clinical trial landscape of various genetic and chronic diseases has been on the rise in recent years. This will help in the growth of the CRISPR gene editing market in the future.
Within the research report, the market is segmented based on product type, application, end user, and region. Each of these segments covers the snapshot of the market over the projected years, the inclination of the market revenue, underlying patterns, and trends by using analytics on the primary and secondary data obtained.
Competitive Landscape
The exponential rise in the application of precision medicine globally has created a buzz among companies to invest in the development of novel CRISPR gene editing. Due to the diverse product portfolio and intense market penetration, Merck KGaA and Thermo Fisher Scientific Inc. have been the pioneers and major competitors in this market. The other major contributors to the market include Integrated DNA Technologies (IDT), Genscript Biotech Corporation, Takara Bio Inc, Agilent Technologies, Inc., and New England Biolabs, Inc.
Based on region, North America holds the largest share of the CRISPR gene editing market due to substantial investments made by biotechnology and pharmaceutical companies, improved healthcare infrastructure, rise in per capita income, early availability of approved therapies, and availability of state-of-the-art research laboratories and institutions in the region. Apart from this, the Asia-Pacific region is anticipated to grow at the fastest CAGR during the forecast period.
Table of Contents
1 Technology Definition
1.1 Inclusion and Exclusion Criteria
1.1.1 Inclusions
1.1.2 Exclusions
2 Research Scope
2.1 Key Questions Answered in the Report
2.2 Target Audience
3 Research Methodology
3.1 CRISPR Gene Editing: Research Methodology
3.2 Primary Data Sources
3.3 Secondary Data Sources
3.4 Market Estimation Model
3.5 Criteria for Company Profiling
4 Market Overview
4.1 Introduction
4.2 CRISPR Gene Editing Market Approach
4.3 Milestones in CRISPR Gene Editing
4.4 CRISPR Gene Editing: Delivery Systems
4.5 CRISPR Technology: A Potential Tool for Gene Editing
4.6 CRISPR Gene Editing Current Scenario
4.7 CRISPR Gene Editing Market: Future Potential Application Areas
5 CRISPR Gene Editing Pipeline Analysis
5.1 CTX001
5.1.1 Product Profile
5.1.2 CTX001 Clinical Trial Status
5.1.2.1 Safety Profile of CTX001
5.2 EDIT-301
5.2.1 Product Profile
5.2.2 EDIT-301 Clinical Trial Status
5.2.2.1 EDIT-301 Preclinical Studies
5.3 NTLA-5001
5.3.1 Product Profile
5.3.1.1 NTLA-5001 Mechanism of Action
5.3.2 NTLA-5001 CLINICAL TRIAL STATUS
5.4 EDIT-101
5.4.1 Product Profile
5.4.2 EDIT-101 CLINICAL TRIAL STATUS
5.4.2.1 Preclinical Studies of EDIT-101
5.4.2.2 Updated Results from Ongoing Phase I/II Clinical Study
5.4.2.3 Preliminary Results of EDIT-101
5.5 OTQ923 and HIX763
5.5.1 Product Profile
5.5.2 OTQ923 and HIX763 CLINICAL TRIAL STATUS
5.6 GPH101
5.6.1 Product Profile
5.6.2 EDIT-101 CLINICAL TRIAL STATUS
5.6.2.1 Efficiency of Targeted Gene Integration
5.6.2.2 Preclinical Validation of GPH101
5.7 BD111
5.7.1 Product Profile
5.7.2 BD111 CLINICAL TRIAL STATUS
5.8 Early Stage Emerging CRISPR Therapies
6 Impact of COVID-19 on Global CRISPR Gene Editing Market
6.1 Impact of COVID-19 on Global CRISPR Gene Editing Market Growth Rate
6.2 Impact on CRISPR Gene Editing Companies
6.3 Clinical Trial Disruptions and Resumptions
6.4 Application of CRISPR Gene Editing in COVID-19
7 Market Dynamics
7.1 Impact Analysis
7.2 Market Drivers
7.2.1 Prevalence of Genetic Disorders and Use of Gene editing
7.2.2 Government and Private Funding
7.2.3 Technology Advancement in CRISPR Gene Editing
7.3 Market Restraints
7.3.1 Off-Target Effects and Delivery
7.3.2 Ethical Concerns and Implications with Respect to Human Gene editing
7.4 Market Opportunities
7.4.1 Expanding Gene and Cell Therapy Area
7.4.2 CRISPR Gene Editing Scope in Agriculture
8 Industry Insights
8.1 Introduction
8.2 Funding Scenario
8.2.1 Funding Scenario: Public Companies
8.2.2 Funding Scenario: Private Companies
8.2.3 Funding Scenario: Government Organizations
8.3 Regulatory Scenario of CRISPR Gene Editing Market
8.3.1 Overview
8.3.2 Legal Requirements and Frameworks in the U.S.
8.3.3 Clinical Trial Authorization
8.3.4 Marketing Authorization
8.3.5 USFDA Guidelines for BLA Submission
8.3.6 Post-Authorization Regulations
8.3.7 Legal Requirements and Frameworks in Europe
8.3.7.1 EMA Biologics License Application Process
8.3.7.2 Centralized Procedure
8.3.7.3 Decentralized Procedure
8.3.7.4 Mutual-Recognition Procedure
8.3.7.5 National Procedure
8.4 Legal Requirements and Frameworks in Asia-Pacific
8.4.1 Legal Requirements and Framework in Japan
8.4.2 Expedited Regulatory Designations around the World
8.5 Pricing of CRISPR Gene Editing
8.6 Reimbursement of CRISPR Gene Editing
8.6.1 CRISPR Gene Editing: Insurance Coverage in the U.S.
9 Global CRISPR Gene Editing Patent Landscape
9.1 Overview
9.2 Global CRISPR Gene Editing Market Patent Landscape (by Application)
9.3 Global CRISPR Gene Editing Market Patent Landscape (by Region)
9.4 CRISPR Gene Editing Market Patent Landscape (by Year)
10 Global CRISPR Gene Editing Market (by Product Type)
10.1 Overview
10.2 CRISPR Products
10.2.1 Kits and Enzymes
10.2.1.1 Vector-Based Cas9
10.2.1.2 DNA-Free Cas9
10.2.2 Libraries
10.2.3 Design Tools
10.2.4 Antibodies
10.2.5 Other Products
10.3 CRISPR Services
10.3.1 gRNA Design and Vector Construction
10.3.2 Cell Line and Engineering
10.3.3 Screening Services
10.3.4 Other Services
11 Global CRISPR Gene Editing Market (by Application)
11.1 Overview
11.2 Agriculture
11.3 Biomedical
11.3.1 Gene Therapy
11.3.2 Drug Discovery
11.3.3 Diagnostics
11.4 Industrial
11.5 Other Applications
12 Global CRISPR Gene Editing Market (by End User)
12.1 Academic Institutes and Research Centers
12.2 Biotechnology Companies
12.3 Contract Research Organizations (CROs)
12.4 Pharmaceutical and Biopharmaceutical Companies
13 Global CRISPR Gene Editing Market (by Region)
13.1 Introduction
13.1.1 Global CRISPR Gene Editing Market (by Region), 2020-2031
13.2 North America
13.2.1 U.S.
13.2.1.1 U.S. CRISPR Gene Editing Market (by Application), 2020-2031
13.2.2 Canada
13.2.2.1 Canada CRISPR Gene Editing Market (by Application), 2020-2031
13.3 Europe
13.3.1 U.K.
13.3.1.1 U.K. CRISPR Gene Editing Market (by Application), 2020-2031
13.3.2 Germany
13.3.2.1 Germany CRISPR Gene Editing Market (by Application), 2020-2031
13.3.3 France
13.3.3.1 France CRISPR Gene Editing Market (by Application), 2020-2031
13.3.4 Spain
13.3.4.1 Spain CRISPR Gene Editing Market (by Application), 2020-2031
13.3.5 Italy
13.3.5.1 Italy CRISPR Gene Editing Market (by Application), 2020-2031
13.3.6 Switzerland
13.3.6.1 Switzerland CRISPR Gene Editing Market (by Application), 2020-2031
13.3.7 Rest-of-Europe
13.3.7.1 Rest-of-Europe CRISPR Gene Editing Market (by Application), 2020-2031
13.4 Asia-Pacific
13.4.1 Japan
13.4.1.1 Japan CRISPR Gene Editing Market (by Application), 2020-2031
13.4.2 China
13.4.2.1 China CRISPR Gene Editing Market (by Application), 2020-2031
13.4.3 South Korea
13.4.3.1 South Korea CRISPR Gene Editing Market (by Application), 2020-2031
13.4.4 Singapore
13.4.4.1 Singapore CRISPR Gene Editing Market (by Application), 2020-2031
13.4.5 Australia
13.4.5.1 Australia CRISPR Gene Editing Market (by Application), 2020-2031
13.4.6 India
13.4.6.1 India CRISPR Gene Editing Market (by Application), 2020-2031
13.4.7 Rest-of-APAC
13.4.7.1 Rest-of-APAC CRISPR Gene Editing Market (by Application), 2020-2031
13.5 Latin America
13.5.1 Brazil
13.5.1.1 Brazil CRISPR Gene Editing Market (by Application), 2020-2031
13.5.2 Mexico
13.5.2.1 Mexico CRISPR Gene Editing Market (by Application), 2020-2031
13.5.3 Rest-of-Latin America
13.5.3.1 RoLA CRISPR Gene Editing Market (by Application), 2020-2031
13.6 Rest-of-the-World
13.6.1 Rest-of-the-World CRISPR Gene Editing Market (by Application), 2020-2031
14 Competitive Landscape
14.1 Key Developments and Strategies
14.1.1 Overview
14.1.1.1 Regulatory and Legal Activities
14.1.1.2 Synergistic Activities
14.1.1.3 Merger and Acquisition Activities
14.1.1.4 Funding Activities
14.2 Market Share Analysis
14.3 Growth Share Analysis
15 Company Profiles
15.1 Overview
15.2 Abcam, Inc.
15.2.1 Company Overview
15.2.2 Role of Abcam, Inc. in the Global CRISPR Gene Editing Market
15.2.3 Financials
15.2.4 SWOT Analysis
15.3 Applied StemCell, Inc.
15.3.1 Company Overview
15.3.2 Role of Applied StemCell, Inc. in the Global CRISPR Gene Editing Market
15.3.3 SWOT Analysis
15.4 Agilent Technologies, Inc.
15.4.1 Company Overview
15.4.2 Role of Agilent Technologies, Inc. in the Global CRISPR Gene Editing Market
15.4.3 Financials
15.4.4 R&D Expenditure
15.4.5 SWOT Analysis
15.5 Cellecta, Inc.
15.5.1 Company Overview
15.5.2 Role of Cellecta, Inc. in the Global CRISPR Gene Editing Market
15.5.3 SWOT Analysis
15.6 CRISPR Therapeutics AG
15.6.1 Company Overview
15.6.2 Role of CRISPR Therapeutics AG in the Global CRISPR Gene Editing Market
15.6.3 Financials
15.6.4 R&D Expenditure
15.6.5 SWOT Analysis
15.7 Thermo Fisher Scientific, Inc.
15.7.1 Company Overview
15.7.2 Role of Thermo Fisher Scientific, Inc. in the Global CRISPR Gene Editing Market
15.7.3 Financials
15.7.4 R&D Expenditure
15.7.5 SWOT Analysis
15.8 GeneCopoeia, Inc.
15.8.1 Company Overview
15.8.2 Role of GeneCopoeia, Inc. in the Global CRISPR Gene Editing Market
15.8.3 SWOT Analysis
15.9 GenScript Biotech Corporation
15.9.1 Company Overview
15.9.2 Role of GenScript Biotech Corporation in the Global CRISPR Gene Editing Market
15.9.3 Financials
15.9.4 SWOT Analysis
15.1 Horizon Discovery Group PLC
15.10.1 Company Overview
15.10.2 Role of Horizon Discovery Group PLC in the Global CRISPR Gene Editing Market
15.10.3 Financials
15.10.4 SWOT Analysis
15.11 Integrated DNA Technologies, Inc.
15.11.1 Company Overview
15.11.2 Role of Integrated DNA Technologies, Inc. in the Global CRISPR Gene Editing Market
15.11.3 SWOT Analysis
15.12 Merck KGaA
15.12.1 Company Overview
15.12.2 Role of Merck KGaA in the Global CRISPR Gene Editing Market
15.12.3 Financials
15.12.4 SWOT Analysis
15.13 New England Biolabs, Inc.
15.13.1 Company Overview
15.13.2 Role of New England Biolabs, Inc. in the Global CRISPR Gene Editing Market
15.13.3 SWOT Analysis
15.14 Origene Technologies, Inc.
15.14.1 Company Overview
15.14.2 Role of Origene Technologies, Inc. in the Global CRISPR Gene Editing Market
15.14.3 SWOT Analysis
15.15 Rockland Immunochemicals, Inc.
15.15.1 Company Overview
15.15.2 Role of Rockland Immunochemicals, Inc. in the Global CRISPR Gene Editing Market
15.15.3 SWOT Analysis
15.16 Synthego Corporation
15.16.1 Company Overview
15.16.2 Role of Synthego Corporation in the Global CRISPR Gene Editing Market
15.16.3 SWOT Analysis
15.17 System Biosciences LLC
15.17.1 Company Overview
15.17.2 Role of System Biosciences LLC in the Global CRISPR Gene Editing Market
15.17.3 SWOT Analysis
15.18 ToolGen, Inc.
15.18.1 Company Overview
15.18.2 Role of ToolGen, Inc. in the Global CRISPR Gene Editing Market
15.18.3 SWOT Analysis
15.19 Takara Bio
15.19.1 Company Overview
15.19.2 Role of Takara Bio in the Global CRISPR Gene Editing Market
15.19.3 Financials
15.19.4 SWOT Analysis
List of Tables
Table 1: CRISPR Technology Vs. Other Gene Editing Technologies
Table 2: Emerging CRISPR Gene Editing Drugs Pipeline
Table 3: Impact of COVID-19 on CRISPR Clinical Trials
Table 4: Private Company Investments in CRISPR Gene Editing
Table 5: Total Funding Received and IPO by CRISPR Gene Editing Public Companies, $Million
Table 6: Funding Scenario of Government Organizations
Table 7: Expedited Regulatory Designations around the World
Table 8: Cost of CRISPR Editing Experiment
Table 9: Cost of CRISPR Gene Editing
List of Figures
Figure 1: Global Total Burden of Diseases by Cause
Figure 2: Global Geriatric Population by Age
Figure 3: CRISPR Gene Editing: Potential Areas of Interest
Figure 4: Global CRISPR Gene Editing Market (by Product Type), $Million, 2020, 2026, and 2031
Figure 5: Global CRISPR Gene Editing: Impact Analysis
Figure 6: Share of Key Developments and Strategies, July 2015-November 2021
Figure 7: Global CRISPR Gene Editing Market (by Product Type), $Million, 2021 and 2031
Figure 8: Global CRISPR Gene Editing Market (by Application), $Million, 2021 and 2031
Figure 9: Global CRISPR Gene Editing Market (by End User), $Million, 2021 and 2031
Figure 10: Global CRISPR Gene Editing Market (by Region), 2021 and 2031
Figure 11: Global CRISPR Gene Editing Market Segmentation
Figure 12: Global CRISPR Gene Editing Market Methodology
Figure 13: Primary Research Methodology
Figure 14: Bottom-Up Approach (Segment-Wise Analysis)
Figure 15: Top-Down Approach (Segment-Wise Analysis)
Figure 16: CRISPR/Cas Systems and Its Classification
Figure 17: CRISPR Gene Editing Approach
Figure 18: Key Milestones in CRISPR Gene Editing
Figure 19: CRISPR Gene Editing Delivery Methods Overview
Figure 20: CRISPR Gene Editing Most Potential Future Application Areas
Figure 21: CRISPR Gene Editing Global Clinical Trials (by Phase), 2021
Figure 22: CRISPR Gene Editing Global Clinical Trials (by Indication), 2021
Figure 23: CRISPR Gene Editing Global Clinical Trials (by Country), 2021
Figure 24: CTX001 Manufacturing and Infusion
Figure 25: CTX001 Phase I/II Clinical Trial Design for Transfusion-Dependent β-Thalassemia
Figure 26: Summary of Adverse Events
Figure 27: EDIT-301-Derived Red Blood Cells Demonstrate Reduced Sickling
Figure 28: EDIT-301 Phase I/II Clinical Trial Design for Sickle Cell Disease
Figure 29: In Vitro and In Vivo Studies for EDIT-101
Figure 30: Advantages of NTLA-5001 for Acute Myeloid Leukaemia (AML)
Figure 31: NTLA-5001: Potential Best-in-Class Engineered T-Cell Therapy for AML
Figure 32: NTLA-5001 Phase I/II Clinical Study
Figure 33: EDIT-101 Mechanism of Action: Removing Mutation with Editing to Correct Vision
Figure 34: EDIT-101 Program Overview
Figure 35: EDIT-101 Phase I/II Clinical Study
Figure 36: EDIT-101 Trial Design
Figure 37: EDIT-101 Safety and Efficacy Tolerability Profile
Figure 38: OTQ923 and HIX763 Mechanism of Action
Figure 39: OTQ923 and HIX763 Phase I/II Clinical Study
Figure 40: GPH101 Mode of Action
Figure 41: GPH101 Phase I/II Clinical Study
Figure 42: GPH101 Phase I/II CEDAR Clinical Trial Design
Figure 43: BD111 Phase I/II Clinical Study
Figure 44: Figure 5.5: Most Impacted Practices in CRISPR Gene Editing
Figure 45: COVID-19 Impact on CRISPR Gene Editing Market
Figure 46: Application of CRISPR/Cas Systems in COVID-19
Figure 47: Global CRISPR Gene Editing: Impact Analysis
Figure 48: Rise in CRISPR PubMed Publications During 2012-2017
Figure 49: NIH Funding for CRISPR-Related Research, $Million, 2015-2019
Figure 50: Technological Advancement Using CRISPR Systems
Figure 51: Major Ethical Concerns of CRISPR Gene Editing
Figure 52: CRISPR Gene Editing Applications in Cell and Gene Therapy
Figure 53: Number of Publications on CRISPR Gene Editing in Plant Gene editing with Agricultural Applications (by Region)
Figure 54: Top Funded CRISPR Gene Editing Public Companies, $Million, 2015-2020
Figure 55: Top-Funded CRISPR Gene Editing Private Companies, $Million
Figure 56: Clinical Trial Authorization for CRISPR Gene Editing
Figure 57: Steps for Obtaining Marketing Authorization
Figure 58: USFDA Review Timeline
Figure 59: EMA Review Timeline
Figure 60: Global CRISPR Gene Editing Market Patents (by Application)
Figure 61: U.S. and China Dominated Patents (by Application)
Figure 62: Global CRISPR Gene Editing Market Patents (by Region)
Figure 63: Global CRISPR Gene Editing Market Patents (by Year)
Figure 64: Revenue Contribution of Different Segments (by Product Type), $Million, 2020 and 2031
Figure 65: Revenue Contribution of CRISPR Products Segments, $Million, 2020 and 2031
Figure 66: Revenue Contribution of Kits and Enzymes Products Segments, $Million, 2020 and 2031
Figure 67: Global CRISPR Gene Editing Market (Kits and Enzymes, by Product), $Million, 2020-2031
Figure 68: Global CRISPR Gene Editing Market (Vector-Based Cas9, by Kits and Enzymes), $Million, 2020-2031
Figure 69: Global CRISPR Gene Editing Market (DNA-Free Cas9, by Kits and Enzymes), $Million, 2020-2031
Figure 70: Global CRISPR Gene Editing Market (Libraries, by Product), $Million, 2020-2031
Figure 71: Global CRISPR Gene Editing Market (Design Tools, by Product), $Million, 2020-2031
Figure 72: Global CRISPR Gene Editing Market (Antibodies, by Product), $Million, 2020-2031
Figure 73: Global CRISPR Gene Editing Market (Other Products, by Product), $Million, 2020-2031
Figure 74: Revenue Contribution of CRISPR Services Segments, $Million, 2020 and 2031
Figure 75: Global CRISPR Gene Editing Market (gRNA Design and Vector Construction, by Services), $Million, 2020-2031
Figure 76: Global CRISPR Gene Editing Market (Cell Line and Engineering, by Services), $Million, 2020-2031
Figure 77: Global CRISPR Gene Editing Market (Screening Services, by Services), $Million, 2020-2031
Figure 78: Global CRISPR Gene Editing Market (Other Services, by Services), $Million, 2020-2031
Figure 79: Revenue Contribution of Different Segments (by Application), $Million 2020 and 2031
Figure 80: CRISPR Gene Editing Market (Agriculture, by Application), $Million, 2020-2031
Figure 81: Revenue Contribution of Different Segments (by Biomedical), $Million, 2020 and 2031
Figure 82: CRISPR Gene Editing Market (Gene Therapy, by Biomedical), $Million, 2020-2031
Figure 83: CRISPR Gene Editing Market (Drug Discovery, by Biomedical), $Million, 2020-2031
Figure 84: CRISPR Gene Editing Market (Diagnostics, by Biomedical), $Million, 2020-2031
Figure 85: CRISPR Gene Editing Market (Industrial, by Application), $Million, 2020-2031
Figure 86: CRISPR Gene Editing Market (Other Applications, by Application), $Million, 2020-2031
Figure 87: Revenue Contribution of Different Segments (by End User), $Million, 2020 and 2031
Figure 88: CRISPR Gene Editing Market (Academic Institutes and Research Centres, by End User), $Million 2020-2031
Figure 89: CRISPR Gene Editing Market (Biotechnology Companies, by End User), $Million, 2020-2031
Figure 90: CRISPR Gene Editing Market (Contract Research Organizations, by End User), $Million, 2020-2031
Figure 91: CRISPR Gene Editing Market (Pharmaceutical and Biopharmaceutical Companies, by End User), $Million, 2020-2031
Figure 92: Global CRISPR Gene Editing Market (by Region)
Figure 93: Global CRISPR Gene Editing Market (by Region), $Million, 2020 and 2031
Figure 94: Global CRISPR Gene Editing Market (by Region), $Million, 2020-2031
Figure 95: North America CRISPR Gene Editing Market, $Million, 2020-2031
Figure 96: North America: Market Dynamics
Figure 97: Revenue Contributions of Various Countries in North America, 2020 and 2031
Figure 98: U.S. CRISPR Gene Editing Market, $Million, 2020-2031
Figure 99: U.S. CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 100: Canada CRISPR Gene Editing Market, $Million, 2020-2031
Figure 101: Canada CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 102: Europe CRISPR Gene Editing Market, $Million, 2020-2031
Figure 103: Europe: Market Dynamics
Figure 104: Revenue Contributions of Various Countries in Europe, $Million, 2020 and 2031
Figure 105: U.K. CRISPR Gene Editing Market, $Million, 2020-2031
Figure 106: U.K. CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 107: Germany CRISPR Gene Editing Market, $Million, 2020-2031
Figure 108: Germany CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 109: France CRISPR Gene Editing Market, $Million, 2020-2031
Figure 110: France CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 111: Spain CRISPR Gene Editing Market, $Million, 2020-2031
Figure 112: Spain CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 113: Italy CRISPR Gene Editing Market, $Million, 2020-2031
Figure 114: Italy CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 115: Switzerland CRISPR Gene Editing Market, $Million, 2020-2031
Figure 116: Switzerland CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 117: Rest-of-Europe CRISPR Gene Editing Market, $Million, 2020-2031
Figure 118: Rest-of-Europe CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 119: Asia-Pacific CRISPR Gene Editing Market, $Million, 2020-2031
Figure 120: Asia-Pacific: Market Dynamics
Figure 121: Revenue Contributions of Various Countries in Asia-Pacific, $Million, 2020 and 2031
Figure 122: Japan CRISPR Gene Editing Market, $Million, 2020-2031
Figure 123: Japan CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 124: China CRISPR Gene Editing Market, $Million, 2020-2031
Figure 125: China CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 126: South Korea CRISPR Gene Editing Market, $Million, 2020-2031
Figure 127: South Korea CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 128: Singapore CRISPR Gene Editing Market, $Million, 2020-2031
Figure 129: Singapore CRISPR gene editing Market (by Application), $Million, 2020-2031
Figure 130: Australia CRISPR Gene Editing Market, $Million, 2020-2031
Figure 131: Australia CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 132: India CRISPR Gene Editing Market, $Million, 2020-2031
Figure 133: India CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 134: Rest-of-APAC CRISPR Gene Editing Market, $Million, 2020-2031
Figure 135: Rest-of-APAC CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 136: Latin America CRISPR Gene Editing Market, $Million, 2020-2031
Figure 137: Latin America: Market Dynamics
Figure 138: Revenue Contributions of Various Countries in Latin America, $Million, 2020 and 2031
Figure 139: Brazil CRISPR Gene Editing Market, $Million, 2020-2031
Figure 140: Brazil CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 141: Mexico CRISPR Gene Editing Market, $Million, 2020-2031
Figure 142: Mexico CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 143: RoLA CRISPR Gene Editing Market, $Million, 2020-2031
Figure 144: RoLA CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 145: Rest-of-the-World CRISPR Gene Editing Market, $Million, 2020-2031
Figure 146: Rest-of-the-World CRISPR Gene Editing Market (by Application), $Million, 2020-2031
Figure 147: Share of Key Developments and Strategies, July 2015-November 2021
Figure 148: Regulatory and Legal Developments Share (by Company), July 2015-November 2021
Figure 149: Synergistic Activities (by Company), July 2015-November 2021
Figure 150: Company Share (by Synergistic Activities), July 2015-November 2021
Figure 151: M&A Activities Share (by Company), July 2015-November 2021
Figure 152: Funding Activities (by Company), July 2015-November 2021
Figure 153: Market Share Analysis (by Company), 2019 and 2020
Figure 154: Growth Share Matrix for Global CRISPR Gene Editing Market (by Company), 2020
Figure 155: Total Number of Companies Profiled
Figure 156: Abcam, Inc.: Overall Product Portfolio
Figure 157: Abcam, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 158: Abcam, Inc.: Overall Financials, $Million, 2018-2020
Figure 159: Abcam, Inc.: Net Revenue (by Region), $Million, 2018-2020
Figure 160: Abcam, Inc.: R&D Expenditure, $Million, 2018-2020
Figure 161: Abcam, Inc.: SWOT Analysis
Figure 162: Applied StemCell, Inc.: Overall Product Portfolio
Figure 163: Applied StemCell, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 164: Applied StemCell, Inc.: SWOT Analysis
Figure 165: Agilent Technologies, Inc.: Overall Product Portfolio
Figure 166: Agilent Technologies, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 167: Agilent Technologies, Inc.: Overall Financials, $Million, 2018-2020
Figure 168: Agilent Technologies, Inc. Net Revenue (by Business Segment), $Million, 2018-2020
Figure 169: Agilent Technologies, Inc.: Revenue (by Region), $Million, 2018-2020
Figure 170: Agilent Technologies, Inc.: R&D Expenditure, $Million, 2018-2020
Figure 171: Agilent Technologies, Inc.: SWOT Analysis
Figure 172: Cellecta, Inc.: Overall Product Portfolio
Figure 173: Cellecta, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 174: Cellecta, Inc.: SWOT Analysis
Figure 175: CRISPR Therapeutics AG: Overall Product Portfolio
Figure 176: CRISPR Therapeutics AG: CRISPR Gene Editing Product Portfolio Pipeline
Figure 177: CRISPR Therapeutics AG: Overall Financials, $Million, 2018-2020
Figure 178: CRISPR Therapeutics AG: R&D Expenditure, $Million, 2018-2020
Figure 179: CRISPR Therapeutics AG: SWOT Analysis
Figure 180: Thermo Fisher Scientific, Inc.: Overall Product Portfolio
Figure 181: Thermo Fisher Scientific, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 182: Thermo Fisher Scientific, Inc.: Overall Financials, $Million, 2018-2020
Figure 183: Thermo Fisher Scientific, Inc.: Net Revenue (by Business Segment), $Million, 2018-2020
Figure 184: Thermo Fisher Scientific, Inc.: Net Revenue (by Region), $Million, 2018-2020
Figure 185: Thermo Fisher Scientific, Inc.: R&D Expenditure, $Million, 2018-2020
Figure 186: Thermo Fisher Scientific, Inc.: SWOT Analysis
Figure 187: GeneCopoeia, Inc.: Overall Product Portfolio
Figure 188: GeneCopoeia, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 189: GeneCopoeia, Inc.: SWOT Analysis
Figure 190: GenScript Biotech Corporation: Overall Product Portfolio
Figure 191: GenScript Biotech Corporation: CRISPR Gene Editing Product Portfolio Category
Figure 192: GenScript Biotech Corporation: Overall Financials, $Million, 2018-2020
Figure 193: GenScript Biotech Corporation: Net Revenue (by Business Segment), $Million, 2018-2020
Figure 194: GenScript Biotech Corporation: Net Revenue (by Region), $Million, 2018-2020
Figure 195: GenScript Biotech Corporation: R&D Expenditure, $Million, 2018-2020
Figure 196: GenScript Biotech Corporation: SWOT Analysis
Figure 197: Horizon Discovery Group PLC: Overall Product Portfolio
Figure 198: Horizon Discovery Group PLC: CRISPR Gene Editing Product Portfolio Category
Figure 199: Horizon Discovery Group PLC: Overall Financials, $Million, 2017-2019
Figure 200: Horizon Discovery Group PLC: Net Revenue (by Business Segment), $Million, 2017-2019
Figure 201: Horizon Discovery Group PLC: Net Revenue (by Region), $Million, 2017-2019
Figure 202: Horizon Discovery Group PLC: R&D Expenditure, $Million, 2017-2019
Figure 203: Horizon Discovery Group PLC: SWOT Analysis
Figure 204: Integrated DNA Technologies, Inc.: Overall Product Portfolio
Figure 205: Integrated DNA Technologies, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 206: Integrated DNA Technologies, Inc.: SWOT Analysis
Figure 207: Merck KGaA: Overall Product Portfolio
Figure 208: Merck KGaA: CRISPR Gene Editing Product Portfolio Category
Figure 209: Merck KGaA: Overall Financials, $Million, 2018-2020
Figure 210: Merck KGaA: Net Revenue (by Business Segment), $Million, 2018-2020
Figure 211: Merck KGaA: Net Revenue (by Region), $Million, 2018-2020
Figure 212: Merck KGaA: R&D Expenditure, $Million, 2018-2020
Figure 213: Merck KGaA: SWOT Analysis
Figure 214: New England Biolabs, Inc.: Overall Product Portfolio
Figure 215: New England Biolabs, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 216: New England Biolabs, Inc.: SWOT Analysis
Figure 217: Origene Technologies, Inc.: Overall Product Portfolio
Figure 218: Origene Technologies, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 219: Origene Technologies, Inc.: SWOT Analysis
Figure 220: Rockland Immunochemicals, Inc.: Overall Product Portfolio
Figure 221: Rockland Immunochemicals, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 222: Rockland Immunochemicals, Inc.: SWOT Analysis
Figure 223: Synthego Corporation: Overall Product Portfolio
Figure 224: Synthego Corporation: CRISPR Gene Editing Product Portfolio Category
Figure 225: Synthego Corporation: SWOT Analysis
Figure 226: System Biosciences LLC: Overall Product Portfolio
Figure 227: System Biosciences LLC: CRISPR Gene Editing Product Portfolio Category
Figure 228: System Biosciences LLC: SWOT Analysis
Figure 229: ToolGen, Inc.: Overall Product Portfolio
Figure 230: ToolGen, Inc.: CRISPR Gene Editing Product Portfolio Category
Figure 231: ToolGen, Inc.: SWOT Analysis
Figure 232: Takara Bio: Overall Product Portfolio
Figure 233: Takara Bio: CRISPR Gene Editing Product Portfolio Category
Figure 234: Takara Bio: Overall Financials, $Million, 2018-2020
Figure 235: Takara Bio: Net Revenue (by Business Segment), $Million, 2018-2020
Figure 236: Takara Bio: Net Revenue (by Region), $Million, 2018-2020
Figure 237: Takara Bio: R&D Expenditure, $Million, 2018-2020
Figure 238: Takara Bio: SWOT Analysis
Companies Mentioned
- Abcam, Inc.
- Applied StemCell, Inc.
- Agilent Technologies, Inc.
- Cellecta, Inc.
- CRISPR Therapeutics AG
- Thermo Fisher Scientific, Inc.
- GeneCopoeia, Inc.
- GenScript Biotech Corporation
- Horizon Discovery Group PLC
- Integrated DNA Technologies, Inc.
- Merck KGaA
- New England Biolabs, Inc.
- Origene Technologies, Inc.
- Rockland Immunochemicals, Inc.
- Synthego Corporation
- System Biosciences LLC
- ToolGen, Inc.
- Takara Bio
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 296 |
| Published | December 2021 |
| Forecast Period | 2021 - 2031 |
| Estimated Market Value ( USD | $ 1445.8 Million |
| Forecasted Market Value ( USD | $ 18856.6 Million |
| Compound Annual Growth Rate | 29.6% |
| Regions Covered | Global |
| No. of Companies Mentioned | 18 |