The COVID-19 pandemic significantly impacted drug manufacturing companies in its preliminary phase. The governments of several countries reported drugs and active pharmaceutical ingredients (APIs) were in short supply due to the outbreak. This was due to cross-border trade barriers that had an ultimatum effect on the import and export of medicinal products, including drugs, resulting in the rise of counterfeit medicines on the market. As per OECD updates from July 2021, the COVID-19 crisis heightened the dangers posed by the global trade in counterfeit pharmaceutical products. Health and safety issues arose when most of the population ordered falsified medicines online since these often needed to be correctly formulated and contain unsafe ingredients. Thus, tackling this global scourge became even more acute and urgent during the pandemic, boosting the use of several counterfeit drug detection devices. Moreover, as per the analysis, this amplified introduction of counterfeit drugs within the market, along with growing volumes of falsified medicines in developing countries due to a lack of stringent regulatory and standardized processes, augmented the demand for counterfeit drug detection over the post-pandemic phase.
Furthermore, the emphasis on advanced surveillance in the supply chain, the shifting trend from packaging security to on-dosage security, and supportive government legislation are some drivers bolstering the market's growth.
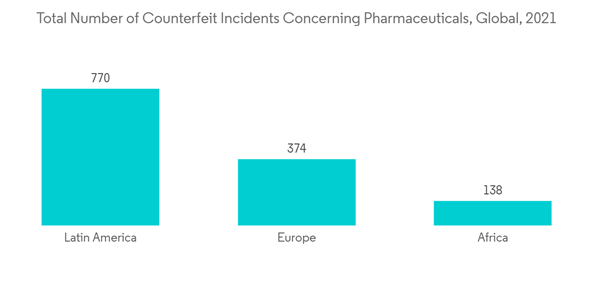
In addition, the rising number of counterfeit drugs is one of the major factors propelling the growth of counterfeit drug detection over the forecast period. Due to the COVID-19 pandemic, counterfeit products increased on the market owing to the crunch in the supply side of drugs. For instance, according to the WHO report, in 2021, the number of cases of substandard and falsified (SF) medical items increased by 52% from 2020. Recently, most incidents involved COVID-19-related medical products, such as vaccines, medicines, COVID test kits, antibiotics, face masks, and sanitizers. In 2021, WHO's surveillance and monitoring system for substandard and counterfeit medical products discovered fraudulent versions of the COVID-19 vaccine COVISHIELD in India and Uganda.
Moreover, a study published in May 2022 in SAGE Journals to describe the law enforcement oversight of falsified drugs by the US FDA from 2016 to 2021 stated that 130 unique enforcement actions were against counterfeiting organizations and individuals. Altogether, 64.6% of enforcement actions implicated counterfeit products marketed and sold over the internet. In 84.6% of activities, counterfeit medicines were sold without a prescription, and in 33.1% activities, the products were marketed as dietary supplements. Sexual dysfunction, anabolic muscle building, benzodiazepines, opioids, stimulants, and dermatologic drugs were the most counterfeited. China was the most predominant country to create counterfeit medicines, followed by India, Turkey, Pakistan, and Russia.
Likewise, pharmaceutical companies are incorporating counterfeit drug detection devices into their supply chains, making them more efficient and enhancing patient safety while saving time and money. For instance, in June 2022, AstraZeneca integrated RFID into its supply chain and had remarkable success dispensing over 30 million RFID-enabled Diprivan syringes.
Thus, these factors mentioned above contribute to the market's growth. However, low awareness about counterfeit drugs will likely limit the market's expansion.
Counterfeit Drug Detection Device Market Trends
RFID Technology Segment Dominates the Market and is Expected to Continue to do so During the Forecast Period
The pharmaceutical industry uses RFID technologies to increase visibility across the value chain and track falsified drugs. The rising number of counterfeit incidents and drug recalls forced the government to reframe drug quality and take strict measures to make QR codes or RFID tags mandatory on active pharmaceutical ingredients and products due to safety and tracking purposes, which are likely to boost the growth of RFID in drug tracing systems in the coming years.As per the report released by ASPA in March 2022, instances of substandard and falsified (SF) medical items surged by about 47% between 2020 and 2021, driven by the COVID-19 pandemic. According to an article published by the NCBI, in 2022, Valsartan was recalled due to identified contamination with N-nitroso dimethylamine (NDMA), a potentially cancerogenic substance, resulting from unintended changes in the manufacturing process in China. Valsartan is a long-term medication in patients with high blood pressure and heart diseases, which causes carcinogenic effects.
Similarly, due to nitrosamine impurities, losartan and hydrochlorothiazide were also recalled from the market. Furthermore, the introduction of next-generation RFID tags and readers is being efficiently used by drug manufacturers and wholesalers in the pharmaceutical industry to prevent medical errors and increase the safety of individualized medicinal products. Thus, the rising number of counterfeit drugs and drug recalls increased the demand for using detection technologies in drug tracing and tracking systems in the pharmaceutical industry. Therefore, the market segment is expected to surge over the forecast period.
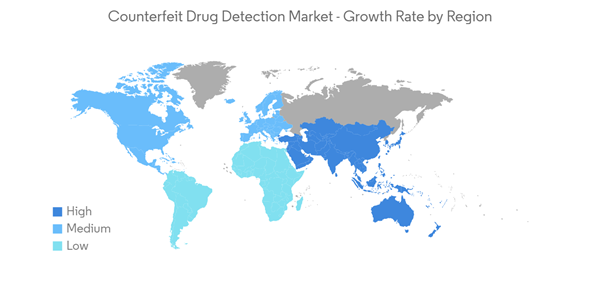
North America is Expected to Hold Significant Market Share in the Forecast Period
North America is anticipated to hold the major market share over the forecast period, owing to the increasing risks from substandard and falsified drugs and stringent regulatory frameworks.The rising incidence of drug counterfeiting in large pharmaceutical and biopharmaceutical companies is the key factor driving the growth of RFID technology in the market. For instance, in December 2020, Janssen Pharmaceutical was notified that counterfeit SYMTUZA had been distributed to over three pharmacies in the United States. The use of RFID is high in supply chain monitoring for tracking assets in real time. Thus, such instances are likely to drive the growth of counterfeit drug detection devices in the pharmaceutical market in the region. Moreover, as per an article published in May 2022 by the Conversation, counterfeiting operations involved tens of millions of pills, more than 1,000 kg (2,200 lbs) of active ingredient powder in the United States, and hundreds of millions of dollars in sales. Unfortunately, with over 11,000 rogue pharmacy sites selling drugs on the internet, these actions barely scratch the surface. The FDA's Office of Criminal Investigations conducts and coordinates criminal investigations into manufacturers and individuals violating federal drug laws. Overall, in 64.6% of cases, the products were sold over the internet, and in 84.6% of the enforcement actions taken, they were obtained without a prescription.
Moreover, the companies developing and launching RFID technologies and systems contribute to the market's growth. For instance, in October 2020, Sandoz partnered with Kit Check, offering artificial intelligence and radio frequency identification medication management technology. Sandoz introduced two RFID-tagged injectable products in the United States under this partnership.
Thus, the market is expected to witness significant growth during the forecast period due to the abovementioned factors.
Counterfeit Drug Detection Device Market Competitor Analysis
The market for counterfeit drug detection is highly fragmented in nature and consists of several global and local players. The major players include Bayer AG, Thermo Fisher Scientific Inc., Spectral Engines Oy, GAO Group, and Systech International. The competitive rivalry is moderate to high due to a small number of regional players. This factor is anticipated to increase the competition for a consolidated customer base. These companies are actively involved in product innovations and technological advancements to sustain their position in the market.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Bayer AG
- Consumer Physics Inc.
- GAO Research Inc.
- Konica Minolta Sensing Americas Inc.
- Rigaku Corporation
- Nynomic Group (Spectral Engines Oy)
- Stratio Inc.
- Systech Group Limited
- Thermo Fisher Scientific Inc.