Clinical Trial Management Systems Market Analysis:
- Major Market Drivers: The clinical trial management systems market growth is fueled by the growing complexity of multisite clinical trials and the gradual adoption of cloud technology. The market size will potentially grow to a considerable extent considering enhanced requirement for trial management modules.
- Key Market Trends: The clinical trial management system outlook showcases trends such as the rise in utilization of decentralized clinical trials, the virtual trial platforms, and risk-based monitoring methods. Moreover, there is an increasing emphasis on interconnectivity and integration with existing trial systems and software platforms.
- Geographical Trends: North America owns the leading position in CTMS market as it has developed advanced healthcare infrastructure and is conducting numerous research and development activities. However, the Asia-Pacific and Latin America regions, which are undergoing rapid market growth because of medical tourism and improving infrastructure, are also offering clinical trial outsourcing services.
- Competitive Landscape: Some of the major market players in the clinical trial management systems industry include Advarra Inc., ArisGlobal LLC, BioClinica Inc., DataTrak International Inc., DZS Clinical Services Inc. (WDB Holdings Co. Ltd.), International Business Machines Corporation, Medidata Solutions Inc. (Dassault Systèmes SE), MedNet Solutions Inc., Oracle Corporation, Parexel International Corporation, RealTime Software Solutions LLC, Veeva Systems Inc., among many others.
- Challenges and Opportunities: The market faces challenges such as data protection issues, interoperability issues and regulatory challenges. It also offers opportunities, such opportunities can be determined from implementation of the advanced analytics, real-world evidence integration, and decentralized trial models.
Clinical Trial Management Systems Market Trends:
Increasing Clinical Trial Complexity
The evolution of precision and personalized medicine has intensified the intricacies of clinical trials, demanding sophisticated management solutions. CTMS platforms play a pivotal role in managing these intricate trials by providing comprehensive solutions for study planning, protocol design, patient enrollment, and data management. By streamlining processes and facilitating collaboration among stakeholders, CTMS platforms help organizations effectively navigate the complexities of modern clinical research, ensuring adherence to rigorous protocols and accelerating the development of innovative therapies. These platforms leverage advanced technologies such as artificial intelligence and machine learning to optimize trial design and execution, enhancing efficiency and generating clinical trial management systems revenue.Stringent Regulatory Requirements
Regulatory bodies impose rigorous guidelines to uphold patient safety and data integrity, necessitating robust compliance management offered by CTMS platforms. CTMS solutions offer robust tools and functionalities to assist organizations in meeting these regulatory obligations. From ensuring protocol adherence and documentation management to facilitating audit readiness and regulatory reporting, CTMS platforms help organizations navigate the complex regulatory landscape with confidence, reducing compliance risks and ensuring adherence to regulatory standards. Additionally, CTMS platforms incorporate features such as automated compliance monitoring and real-time risk assessment to proactively identify and address regulatory issues, enhancing overall compliance efficiency and effectiveness. For instance, the regulations for clinical trials in the United States are governed by stringent guidelines such as the Food and Drug Omnibus Reform Act of 2022 (FDORA), Privacy Act of 1974, and the NIH Policy Manual on Privacy and Confidentiality.Emphasis on Patient-Centric Trials
With a shift toward patient-centricity, CTMS platforms prioritize features like patient recruitment portals and remote monitoring to enhance patient engagement and trial success. There is a growing recognition of the importance of patient-centricity in clinical research, with a focus on enhancing patient engagement, experience, and retention. CTMS platforms support this paradigm shift by incorporating features such as patient recruitment portals, remote monitoring capabilities, and electronic patient-reported outcomes (ePRO). By empowering patients with tools for active participation and communication, CTMS platforms improve patient compliance, enhance data quality, and ultimately contribute to the success of patient-centric trials. This is creating a positive clinical trial management systems market overview.Clinical Trial Management Systems Market Segmentation:
This report provides an analysis of the key trends in each segment of the market, along with forecasts at the global, regional, and country levels for 2025-2033. The report categorizes the market based on component, deployment mode, and end user.Breakup by Component:
- Software
- Services
Software accounts for the majority of the market share
The report has provided a detailed breakup and analysis of the market based on the component. This includes software, and services. According to the report, software represented the largest segment.The expanding penetration of software in the CTMS market is mainly induced by the potential for added flexibility, scalability, and customization. Software offerings provide bespoke functions for trial design, subject recruitment, data management, and regulatory conformity as clinical trial ecosystem has different stakeholders. Apart from this, SaaS models facilitate in having cost-effective deployment and easy accessibility, especially for the small and mid-sized organizations. The growing sophistication of the clinical trials and implicit need for advanced analytics and real-time insights has placed software at the heart of trial workflow management, process improvement, and drug development speedup. For instance, the U.S. Federal Government, through the Department of Health and Human Services (HHS) and the National Institutes of Health (NIH), has proposed policy changes to improve clinical trial transparency by expanding trial registration requirements and data sharing to enhance research transparency.
Breakup by Deployment Mode:
- Web-based CTMS
- On-premise
- Cloud-based CTMS
Web-based CTMS holds the largest share of the industry
A detailed breakup and analysis of the market based on the deployment mode have also been provided in the report. This includes web-based CTMS, on-premise, and cloud-based CTMS. According to the report, web-based CTMS accounted for the largest market share.The web-based CTMS as a deployment mode dominates the CTMS market as web-based methods provide more availability and convenience as users can access the system from any location worldwide where there is internet connection. This makes it possible for a stakeholder team to work remotely and have ready data access, ensuring the smooth running of trials. Moreover, online CTMS systems typically have lower initial costs and fewer IT infrastructure investments than on-site installations making them to be terrific options to small and mid-sized organization looking for an economical and scalable trials management solutions. For instance, Sitero recently acquired the Clario eClinical suite including, Mentor CTMS, a web-based CTMS solution offer benefits, such as easy and affordable third-party eTMF integration, unrestricted access, a familiar Microsoft Office-based interface, rapid implementation, and mobility-enabled features, catering to the demand for efficient and user-friendly CTMS platforms.
Breakup by End User:
- Pharmaceutical and Biotechnology Firms
- Contract Research Organizations
- Others
Pharmaceutical and biotechnology firms represent the leading market segment
The report has provided a detailed breakup and analysis of the market based on the end user. This includes pharmaceutical and biotechnology firms, contract research organizations, and others. according to the report, pharmaceutical and biotechnology firms represented the largest market share.Clinical trials are becoming more complex and highly scrutinized making it necessary for pharmaceuticals and biotechnology companies to incorporate CTMS solutions on a daily basis as the end users. CTMS platforms furnish with all-encompassing features that are meant to be customized for the use of the pharmaceutical and biotech companies, and hence trials operations can be seamlessly carried out. The collaboration of different stakeholders is enhanced and, besides, regulatory compliance is maintained, and therapies developed much faster. Furthermore, CTMS systems are useful to these entities in reducing resources utilization, controlling the clinical trial costs and improving the overall organization productivity, thus improving the clinical trial management systems market statistics.
Breakup by Regional:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America leads the market, accounting for the largest clinical trial management systems market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America holds the leading position in the market for clinical trial management systems.North America drives the CTMS market due to its robust healthcare infrastructure, extensive clinical research activities, and stringent regulatory standards. The region hosts numerous pharmaceutical, biotechnology, and medical device companies conducting clinical trials, creating a significant demand for CTMS solutions. Additionally, the prevalence of chronic diseases and the need for innovative therapies further fuel the adoption of advanced trial management tools. Moreover, North America's substantial investments in research and development, coupled with its technological advancements and focus on precision medicine, contributing to the the CTMS market growth.
Competitive Landscape:
- The market research report has also provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the major market players in the clinical trial management systems industry include Advarra Inc., ArisGlobal LLC, BioClinica Inc., DataTrak International Inc., DZS Clinical Services Inc. (WDB Holdings Co. Ltd.), International Business Machines Corporation, Medidata Solutions Inc. (Dassault Systèmes SE), MedNet Solutions Inc., Oracle Corporation, Parexel International Corporation, RealTime Software Solutions LLC and Veeva Systems Inc.
- Key players in the CTMS market are focusing on innovation and strategic partnerships to enhance their offerings and expand their market presence. They are investing in advanced technologies such as artificial intelligence and machine learning to improve trial efficiency and data analytics capabilities, thereby providing the global clinical trial management systems market opportunities. Additionally, these companies are collaborating with pharmaceutical firms, contract research organizations, and academic institutions to develop integrated solutions that address the evolving needs of clinical research. Moreover, they are expanding their global footprint through acquisitions and regional expansions to tap into emerging markets and capitalize on the growing demand for CTMS solutions worldwide. For instance, On 27 September 2023, Emmes, known for its clinical research services and technology solutions, is now utilizing telehealth features of its advantage eClinical platform. Emmes' move of combining these productivity and operating tools under a single strategic plan confirms its commitment to delivering more integrated services and meeting the requirements of the continuously shifting terrain in clinical research.
Key Questions Answered in This Report
1. What was the size of the global clinical trial management systems market in 2024?2. What is the expected growth rate of the global clinical trial management systems market during 2025-2033?
3. What has been the impact of COVID-19 on the global clinical trial management systems market?
4. What are the key factors driving the global clinical trial management systems market?
5. What is the breakup of the global clinical trial management systems market based on the component?
6. What is the breakup of the global clinical trial management systems market based on the deployment mode?
7. What is the breakup of the global clinical trial management systems market based on the end user?
8. What are the key regions in the global clinical trial management systems market?
9. Who are the key players/companies in the global clinical trial management systems market?
Table of Contents
Companies Mentioned
- Advarra Inc.
- ArisGlobal LLC
- BioClinica Inc.
- DataTrak International Inc.
- DZS Clinical Services Inc. (WDB Holdings Co. Ltd.)
- International Business Machines Corporation
- Medidata Solutions Inc. (Dassault Systèmes SE)
- MedNet Solutions Inc.
- Oracle Corporation
- Parexel International Corporation
- RealTime Software Solutions LLC
- Veeva Systems Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 139 |
| Published | May 2025 |
| Forecast Period | 2024 - 2033 |
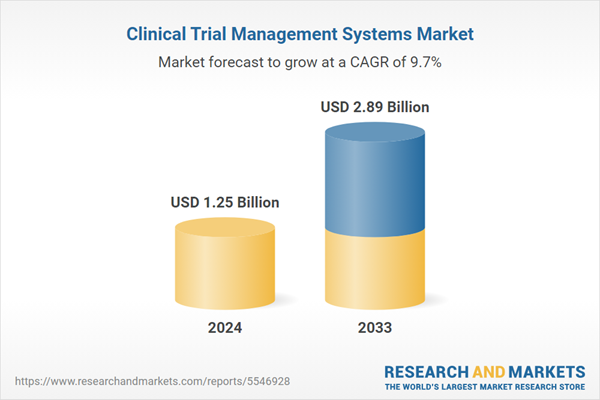
| Estimated Market Value ( USD | $ 1.25 Billion |
| Forecasted Market Value ( USD | $ 2.89 Billion |
| Compound Annual Growth Rate | 9.7% |
| Regions Covered | Global |
| No. of Companies Mentioned | 12 |