Global Generic Injectables Market Overview
Generic injectables are similar to their brand-name counterparts in terms of active ingredients, clinical performance, and intended use, among others. They are administered generally through intramuscular or intravenous routes. Generic injectables offer a faster therapeutic effect, higher bioavailability, and can be formulated for slow and sustained release. The rising patent expiration of branded drugs is boosting the market entry of their generic versions, which are typically lower in price and show similar efficacy and safety profiles. The increasing prevalence of chronic diseases and the growing aging population significantly contribute to the rising market demand for generic injectables. Further, supportive policies and initiatives by the government also fuel the market growth by encouraging the use of generic drugs and ensuring faster approval processes for first-time generics.Global Generic Injectables Market Growth Drivers
Surge in Drug Approvals of Generic Injectables to Affect the Market Landscape Significantly
The market is witnessing a surge in drug approvals of generic injectables by health regulatory agencies such as the United States Food and Drug Corporation (FDA). For instance, in April 2024, an India-based generic injectable manufacturer Gland Pharma Limited received FDA approval for its generic Eribulin Mesylate Injection (0.5 mg/mL single-dose vial) to treat breast cancer. The product, which Gland Pharma co-developed with a research-oriented pharmaceutical product development company Orbicular Pharmaceutical Technologies Private Ltd, is expected to be the first generic intravenous drug for this condition in the United States. The rise in approval of new generic injectables, accelerated by the presence of a supportive regulatory framework, is anticipated to influence the market dynamics.Launch of New Products to Meet Rising Generic Injectables Market Demand
In March 2024, Meitheal Pharmaceuticals, Inc., a generic injectables company based in Chicago, announced recent business progress to support a sustainable drug supply in the United States following its generic injectables portfolio expansion in September 2023. The company is preparing to launch up to 20 new products in 2024 spread across various therapeutic areas and has invested over USD 100 million in generic research and development over the past six years. Additionally, the generic injectables manufacturer recently launched multiple presentations of bupivacaine hydrochloride injection (an anesthetic used for surgical, therapeutic, diagnostic, and obstetrical procedures). The rising introduction of sustainably manufactured and priced generic injectables is poised to augment market demand.Global Generic Injectables Market Trends
The market is witnessing several trends and developments to improve the current global scenario. Some of the notable trends are as follows:Rising Technological Advancements in Manufacturing Processes
The rising advancements in technology are allowing manufacturers to improve the efficiency and scalability of generic injectable production. Integration of innovations such as automation and data analytics in manufacturing processes helps in reduce costs and enhance quality control. This leads to the development of more reliable and consistent products, enabling companies to meet stringent regulatory standards and contributing to the overall market growth.Increased Strategic Mergers and Acquisitions
Recent market trends reveal consolidation via mergers and acquisitions among the key companies to boost market share and expand product portfolios. This strategy enables companies to enter new markets, optimize their supply chains, and increase R&D capabilities by sharing resources and expertise. Such strategic partnerships drive innovation and also elevate the generic injectables market value.Growing Focus on Sterile Injectables
The increasing focus on sterile injectables is in response to the need for higher safety standards in medication administration. This trend has led to substantial investments in cleanroom technology and quality control systems. The growing emphasis on sterile injectables is likely to improve patient safety and increase the demand for generic injectables in clinical settings.Expansion into Emerging Markets
Many generic injectables manufacturers are increasingly expanding their presence into emerging markets such as Asia, Latin America, and Africa. These regions offer significant growth opportunities due to rising healthcare needs and increasing investment in healthcare infrastructure. The rising introduction of international companies in these emerging economies is poised to increase access to affordable injectables in underserved areas, thereby contributing to improved patient outcomes.Global Generic Injectables Market Segmentation
“Generic Injectables Market Report and Forecast 2025-2034” offers a detailed analysis of the market based on the following segments:Market Breakup by Product Type
- Large Molecule Injectables
- Monoclonal Antibodies (mAbs)
- Insulin
- Others
- Small Molecule Injectables
Market Breakup by Container Type
- Vials
- Premix
- Prefilled Syringes
- Ampoules
- Others
Market Breakup by Application
- Oncology
- Cardiovascular
- CNS
- Infectious Diseases
- Autoimmune Disorders
- Others
Market Breakup by Route of Administration
- Intravenous
- Intramuscular
- Subcutaneous
- Others
Market Breakup by Distribution Channel
- Hospital
- Retail Pharmacy
- Others
Market Breakup by Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
Global Generic Injectables Market Share
Oncology Segment Leads Market Share by Application
Based on the application, the market is segmented into oncology, cardiovascular, CNS, infectious diseases, and autoimmune disorders, among others. The oncology segment covers a significant market share due to the rising burden of cancer, which increases the demand for chemotherapy treatments that often require generic injectables. Generic carboplatin and cisplatin are platinum-based, injectable chemotherapeutic drugs that are commonly used in curative regimens and combination therapies. Further, cardiovascular and Infectious segments also hold a high market value owing to its efficacy and rapid onset of action of various generic injectables in treating cardiovascular conditions and infectious diseases.Market Segmentation Based on Container Type is Anticipated to Witness Substantial Growth
Container types available in the market include vials, premix, prefilled syringes, and ampoules, among others. The market share is driven by the rising need for reliable and cost-effective containers that are compatible with a wide range of drug formulations. Vial is the traditional container type and is commonly used due to its affordability, particularly for high-volume production. Additionally, the ease of manufacture and the simplicity of design make it a preferred choice for many pharmaceutical companies.Global Generic Injectables Market Analysis by Region
Based on the region, the market covers North America, Europe, Asia Pacific, Latin America, Middle East and Africa, with each region contributing to the overall dynamics of the market. North America holds the largest market share, valued at USD 33 billion in 2023, which can be attributed to the increasing burden of chronic diseases and the growing aging population. For instance, the American Cancer Society's annual cancer statistics report estimated that new cancer cases will surpass 2 million in the United States in 2024, which indicates the need for effective and affordable generic drugs including generic injectables.Europe also holds a high market value, with the market size estimated to be USD 13.5 billion in 2023, owing to the rising healthcare needs and the presence of a favorable regulatory environment. Asia Pacific is projected to witness exponential growth driven by increased government initiatives that promote the uptake and manufacture of generic injectables.
Leading Players in the Global Generic Injectables Market
The key features of the market report include patent analysis, grants analysis, funding and investment analysis, partnerships, and collaborations analysis by the leading key players. The major companies in the market are as follows:Pfizer Inc.
The global pharmaceutical giant Pfizer has a strong presence in the generic injectables market. Acquired in 2015, its subsidiary Hospira specializes in injectable drugs and infusion technologies.Teva Pharmaceutical Industries Ltd.
Headquartered in Israel, Teva is one of the largest generic drug manufacturers in the world. The company offers a range of generic injectable products in multiple therapeutic areas including pain management and oncology.Baxter
Baxter International Inc. is a global leader in generic injectables, specializing in anesthesia and critical care domain. The company is popular for its innovative delivery systems and premixed injectable drugs.Novartis Pharmaceuticals Corporation
In October 2023, Novartis completed the Spin-off of its generics and biosimilars business, Sandoz, which is a key player in the generic injectables market, focusing on therapeutic areas such as oncology, immunology, and endocrinology.Other players in the market include Fresenius SE & Co. KGaA, Endo, Inc., Hikma Pharmaceuticals PLC, Dr. Reddy's Laboratories Ltd., Sagent Pharmaceuticals, Viatris Inc. (Mylan N.V.), Biocon, Sanofi, Lupin, Aurobindo Pharma Limited, and Sun Pharmaceutical Industries Ltd, among others.
Key Questions Answered in the Global Generic Injectables Market Report
- What was the global generic injectables market value in 2024?
- What is the global generic injectables market forecast outlook for 2025-2034?
- What are the regional markets covered in the report?
- What is market segmentation based on product type?
- What is the market breakup based on container type?
- What are the major applications areas of generic injectables?
- What is the market breakup by route of administration?
- What are the distribution channels in the market?
- What are the major factors aiding the global generic injectables market demand?
- How has the market performed so far and how is it anticipated to perform in the coming years?
- What are the market's major drivers, opportunities, and restraints?
- Which regional market is expected to lead the market share in the forecast period?
- Which country is expected to experience expedited growth during the forecast period?
- How do the prevalence and incidence of chronic diseases affect the market landscape?
- What are the major global generic injectables market trends?
- How does the rise in the geriatric population impact the market size?
- Which product type will dominate the market share?
- Which application area is expected to have a high market value in the coming years?
- Which container type will experience the highest demand in the market segment?
- Which distribution channel will contribute significantly to the market growth?
- Which route of administration is poised to lead the market share?
- Who are the key players involved in the generic injectables market?
- What is the patent landscape of the market?
- What are the current unmet needs and challenges in the market?
- How are partnerships, collaborations, mergers and acquisitions among the key market players shaping the market dynamics?
This product will be delivered within 3-5 business days.
Table of Contents
Companies Mentioned
- Pfizer Inc.
- Teva Pharmaceutical Industries Ltd.
- Baxter.
- Novartis Pharmaceuticals Corporation
- Fresenius SE & Co. KGaA
- Endo, Inc.
- Hikma Pharmaceuticals PLC
- Dr.Reddy’s Laboratories Ltd.
- Sagent Pharmaceuticals
- Viatris Inc.
- Biocon
- Sanofi
- Lupin.
- Aurobindo Pharma Limited
- Sun Pharmaceutical Industries Ltd.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 350 |
| Published | July 2025 |
| Forecast Period | 2025 - 2034 |
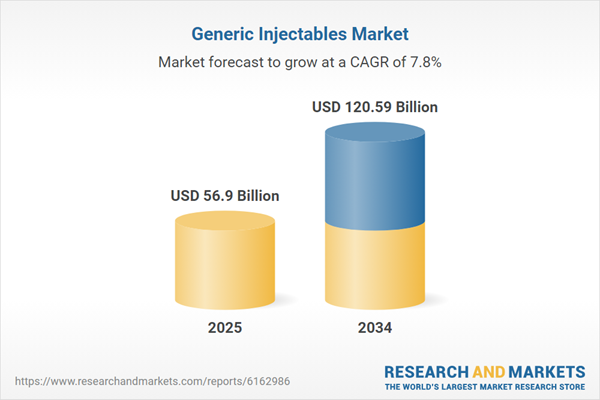
| Estimated Market Value ( USD | $ 56.9 Billion |
| Forecasted Market Value ( USD | $ 120.59 Billion |
| Compound Annual Growth Rate | 7.8% |
| Regions Covered | Global |
| No. of Companies Mentioned | 15 |