Immunoglobulins are the antibodies produced naturally by the body’s immune system, which help fight infection and disease. Individuals with immunoglobulin deficiency require the external administration of immunoglobulin and known as immunoglobulin replacement therapy (IgRT). IgRT can be given intravenously and subcutaneously. Intravenous immunoglobulin (IVIg) and subcutaneous immunoglobulin (SCIg) are selected based on the indication and severity. IVIgs are extracted from human plasma. These antibodies are used in the treatment of various immunological, hematological, and neurological illnesses. Factors such as rising immunodeficiency disease prevalence and increasing geriatric population in the US drive the US intravenous immunoglobulin market growth. However, adverse effects of immunoglobulin therapy hamper the market growth.
IVIGs are used to treat patients suffering from primary and secondary immunodeficiencies and autoimmune disorders. The IVIG is a standard therapy to treat most primary immunodeficiencies (PIDs). According to data by John Hopkins Medicine, ~3% of the total US population, i.e., 10 million people, suffers from autoimmune diseases. As per the National Institute of Allergy and Infectious Diseases (NIAID), there are over 200 different types of primary immune deficiency diseases (PIDDs) affecting ~500,000 people in the US. The rare genetic diseases can be chronic, weakening, and expensive to treat. Immune Thrombocytopenia (ITP) is an autoimmune bleeding disorder characterized by abnormally low levels of platelets; a situation referred to as thrombocytopenia. The Rare Diseases Organization states that the incidence rate of ITP in adults is ~3.3 per 100,000 per year in the US. Per the National Multiple Sclerosis Society study report, published in 2019, ~1 million adults (~913,925) were affected by multiple sclerosis (MS) in the US. According to the MS Discovery Forum, ~200 new cases are diagnosed each week in the US. Thus, the increasing prevalence of immunodeficiency diseases drives the growth of the US intravenous immunoglobulin market.
Many market players and research institutes operating in the US intravenous immunoglobulin market are developing advanced technologies to expand their product portfolios and increase their market shares. Rising immunodeficiency disease prevalence, product approvals, and collaborations are among the factors driving the US intravenous immunoglobulin market size.
A few of the recent developments related to US intravenous immunoglobulin are mentioned below:
In April 2020, Kamada Ltd. and Kedrion Biopharma collaborated to develop, manufacture, and distribute a human plasma-derived Anti-SARS-CoV-2 (COVID-19) polyclonal immunoglobulin (IgG) product as a potential treatment against COVID-19. The announcement follows separate previous reports by the two entities concerning initiating development programs for such products.
In January 2022, LFB and Kedrion joined forces in an industrial cooperation agreement to increase the availability of immunoglobulins for patients in France. In the framework of this industrial partnership, Kedrion manufactured immunoglobulins in Hungary from LFB plasma which was collected in France by the “Etablissement Français du Sang” (EFS). LFB imported these immunoglobulins manufactured by Kedrion in France and complemented its immunoglobulins, which are available in hospitals across France, in agreement with and under the control of French health authorities.
In April 2020, In April 2020, Octapharma collaborated with CSL, Takeda, BPL, Biotest, and LFB to investigate the possibility of treating COVID-19 with plasma-derived hyperimmune immunoglobulins.
US Intravenous Immunoglobulin Market, By Type-Based Insights
Based on product type, the US intravenous immunoglobulin market is segmented into IgG, IgM, IgA, and others. In 2022, the IgG segment held the largest share of the market. Moreover, the same segment is expected to record the highest CAGR during the forecast period.IVIg is a replacement therapy and treatment for patients with antibody deficiencies or for those who suffer from immunodeficiency disorders. For instance, in primary or secondary hypogammaglobulinemia, IVIg replacement therapy provides protection against infections by administering an adequate volume of IgG in the blood. IVIg is a blood product prepared from serum derived from 1,000-15,000 donors per batch where only IgG is purified from plasma to produce IVIg products. Therefore, IVIg used for treatment therapies contains 95-98% pure IgG with small amounts of other plasma proteins, including IgA and IgM.
Thus, wide applications of IgG in treating several categories of disorders are among the factors contributing to the growth of the US intravenous immunoglobulin market.
US Intravenous Immunoglobulin Market, By End User-Based Insights
The US intravenous immunoglobulin market, based on end user, is segmented into hypogammaglobulinemia, chronic inflammatory demyelinating polyneuropathy (CIDP), immunodeficiency diseases, myasthenia gravis (MG), multifocal motor neuropathy (MMN), idiopathic thrombocytopenic purpura (ITP), inflammatory myopathies, specific antibody deficiency (SDA), Guillain-Barre syndrome (GBS), and others. In 2022, the immunodeficiency diseases segment held the largest market share in . The same segment is expected to grow at the fastest rate during the coming years.The World Health Organization (WHO), the US Census Bureau, and the US National Library of Medicine are a few of the major primary and secondary sources referred to while preparing the report on the US intravenous immunoglobulin market.
Table of Contents
Companies Mentioned
- Takeda Pharmaceuticals Co Ltd
- Grifols Sa
- Pfizer Inc
- Adma Biologics
- Bio Products Laboratory Ltd
- Octapharma AG
- Kedrion Spa
- Csl Ltd
- Lfb Group
- Prothya Biosolutions Bv
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 145 |
| Published | April 2023 |
| Forecast Period | 2023 - 2028 |
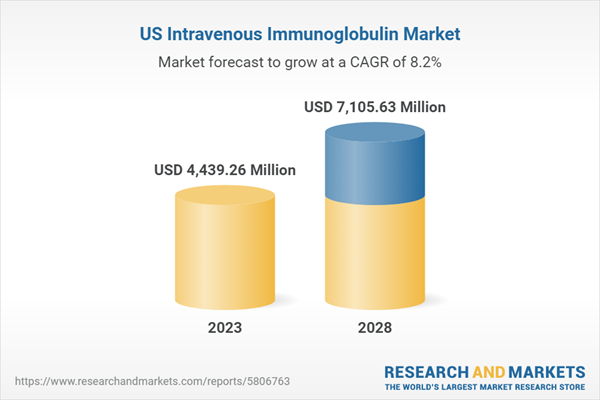
| Estimated Market Value ( USD | $ 4439.26 Million |
| Forecasted Market Value ( USD | $ 7105.63 Million |
| Compound Annual Growth Rate | 8.2% |
| Regions Covered | United States |
| No. of Companies Mentioned | 10 |