Semi-automated Automated External Defibrillators (AEDs) represent a vital component of emergency medical response systems, offering a balanced approach between user intervention and automated assistance. These devices are equipped with features that require users to manually initiate the delivery of an electric shock following the analysis of the victim's heart rhythm. Therefore, the UK market consumed 152.1 hundred units Semi-automated AEDs in the market in 2022.
The Germany market dominated the Europe Automated External Defibrillator Market by Country in 2022, and would continue to be a dominant market till 2030; thereby, achieving a market value of $181.7 million by 2030. The UK market is exhibiting a CAGR of 8.2% during (2023 - 2030). Additionally, The France market would experience CAGR of 10% during (2023 - 2030).
AED adoption is further facilitated by integration with emergency services. Many communities have established AED registries or databases that help emergency responders locate nearby AEDs quickly. Additionally, advancements in connectivity allow AEDs to transmit data in real-time to emergency services, enabling a more coordinated and immediate response to cardiac emergencies. For instance, according to statistics from the British Heart Foundation, by 2024, around 620 million individuals suffer from heart and circulatory diseases.
Many workplaces, especially those with higher risks of cardiac events, have adopted AEDs as part of their safety measures. Factories and industrial facilities deploy AEDs to address cardiac emergencies in potentially hazardous environments. AEDs are present on construction sites where immediate medical assistance may be critical. AEDs are part of resuscitation equipment, supporting medical professionals in emergencies.
As per European Cardiovascular Disease Statistics 2017, over 1.8 million deaths in the European Union and 3.9 million in Europe are attributed to cardiovascular disease (CVD) each year. In Europe and the EU, CVD is the primary cause of mortality in 45% and 37% of cases, respectively. Europe has a significant prevalence of risk factors for cardiovascular diseases, such as hypertension, diabetes, and lifestyle-related factors. Thus, all these factors will uplift the regional market’s expansion in the coming years.
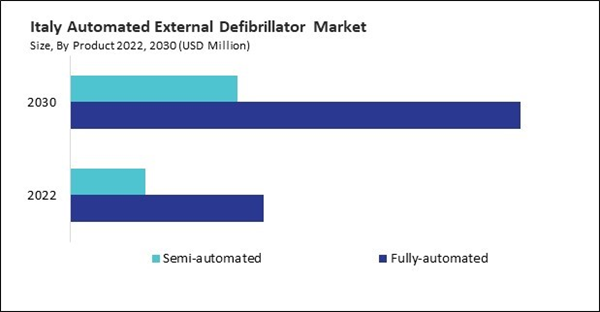
Based on Product, the market is segmented into Fully-automated, and Semi-automated. Based on End-use, the market is segmented into Hospital, Pre Hospital, Public Access, Homecare, and Others. Based on countries, the market is segmented into Germany, UK, France, Russia, Spain, Italy, and Rest of Europe.
List of Key Companies Profiled
- Nihon Kohden Corporation (Defibtech LLC)
- Koninklijke Philips N.V.
- Bpl Medical Technologies Private Limited
- Corpuls (Nordic Capital)
- Progetti SRL
- Stryker Corporation (HeartSine Technologies, Ltd.)
- CU Medical Systems, Inc.
- Silverline Meditech Pvt. Ltd.
- AMI Italia S.r.l.
- Medtronic PLC
Market Report Segmentation
By Product (Volume, Hundred Units, USD Billion, 2019-2030)- Fully-automated
- Semi-automated
- Hospital
- Pre Hospital
- Public Access
- Homecare
- Others
- Germany
- UK
- France
- Russia
- Spain
- Italy
- Rest of Europe
Table of Contents
Companies Mentioned
- Nihon Kohden Corporation (Defibtech LLC)
- Koninklijke Philips N.V.
- Bpl Medical Technologies Private Limited
- Corpuls (Nordic Capital)
- Progetti SRL
- Stryker Corporation (HeartSine Technologies, Ltd.)
- CU Medical Systems, Inc.
- Silverline Meditech Pvt. Ltd.
- AMI Italia S.r.l.
- Medtronic PLC