The anaplastic astrocytoma market has been comprehensively analyzed in this report titled "Anaplastic Astrocytoma Market: Epidemiology, Industry Trends, Share, Size, Growth, Opportunity, and Forecast 2024-2034". Anaplastic astrocytoma is a type of brain tumor that arises from astrocytes, which are star-shaped cells that provide support and nourishment to nerve cells in the brain. The disease is mainly characterized by the presence of rapidly dividing, abnormal astrocytic cells. The symptoms of this ailment can vary depending on the location and size of the tumor. Individuals suffering from the condition may experience persistent headaches, double or blurred vision, seizures, changes in behavior or personality, cognitive impairments, focal neurological deficits, etc. The diagnosis of anaplastic astrocytoma typically involves a combination of underlying indications, medical history, and neurological studies. Several other diagnostic procedures, like magnetic resonance imaging (MRI), immunohistochemistry, fluorescence in-situ hybridization analysis, etc., are also utilized to help in identifying the presence of specific markers that are characteristic of this condition. The healthcare provider may further perform a histological examination of a tissue sample obtained through a brain biopsy to determine the grade and type of tumor cells among patients.
The increasing cases of genetic disorders that cause numerous gene mutations in cellular DNA are primarily driving the anaplastic astrocytoma market. In addition to this, the rising incidences of various risk factors, such as prior exposure to high-dose radiation, advancing age, unhealthy diet, family history of gliomas, stress, etc., are also bolstering the market growth. Furthermore, the widespread adoption of anti-angiogenic therapy, which aims to inhibit the development of new blood vessels that supply nutrients to the cancerous cells, thereby preventing disease progression, is acting as another significant growth-inducing factor. Additionally, the escalating demand for tumor-treating field treatment, which uses low-intensity or frequency-alternating electrical fields to disrupt cell division and block the proliferation of tumors in patients, is also creating a positive outlook for the market. Apart from this, the emerging popularity of targeted therapies, since they aim to directly interfere with specific molecules or signaling pathways that are involved in the survival of cancerous cells while potentially sparing the surrounding healthy tissues, is expected to drive the anaplastic astrocytoma market in the coming years.
This report provides an exhaustive analysis of the anaplastic astrocytoma market in the United States, EU5 (Germany, Spain, Italy, France, and United Kingdom) and Japan. This includes treatment practices, in-market, and pipeline drugs, share of individual therapies, market performance across the seven major markets, market performance of key companies and their drugs, etc. The report also provides the current and future patient pool across the seven major markets. According to the report the United States has the largest patient pool for anaplastic astrocytoma and also represents the largest market for its treatment. Furthermore, the current treatment practice/algorithm, market drivers, challenges, opportunities, reimbursement scenario and unmet medical needs, etc. have also been provided in the report. This report is a must-read for manufacturers, investors, business strategists, researchers, consultants, and all those who have any kind of stake or are planning to foray into the anaplastic astrocytoma market in any manner.
Time Period of the Study
- Base Year: 2023
- Historical Period: 2018-2023
- Market Forecast: 2024-2034
Countries Covered
- United States
- Germany
- France
- United Kingdom
- Italy
- Spain
- Japan
Analysis Covered Across Each Country
- Historical, current, and future epidemiology scenario
- Historical, current, and future performance of the anaplastic astrocytoma market
- Historical, current, and future performance of various therapeutic categories in the market
- Sales of various drugs across the anaplastic astrocytoma market
Competitive Landscape:
This report also provides a detailed analysis of the current anaplastic astrocytoma marketed drugs and late-stage pipeline drugs.In-Market Drugs
- Drug Overview
- Mechanism of Action
- Regulatory Status
- Clinical Trial Results
- Drug Uptake and Market Performance
Late-Stage Pipeline Drugs
- Drug Overview
- Mechanism of Action
- Regulatory Status
- Clinical Trial Results
- Drug Uptake and Market Performance
Key Questions Answered in this Report:
Market Insights
- How has the anaplastic astrocytoma market performed so far and how will it perform in the coming years?
- What are the markets shares of various therapeutic segments in 2023 and how are they expected to perform till 2034?
- What was the country-wise size of the anaplastic astrocytoma market across the seven major markets in 2023 and what will it look like in 2034?
- What is the growth rate of the anaplastic astrocytoma market across the seven major markets and what will be the expected growth over the next ten years?
- What are the key unmet needs in the market?
Epidemiology Insights
- What is the number of prevalent cases (2018-2034) of anaplastic astrocytoma across the seven major markets?
- What is the number of prevalent cases (2018-2034) of anaplastic astrocytoma by age across the seven major markets?
- What is the number of prevalent cases (2018-2034) of anaplastic astrocytoma by gender across the seven major markets?
- How many patients are diagnosed (2018-2034) with anaplastic astrocytoma across the seven major markets?
- What is the size of the anaplastic astrocytoma patient pool (2018-2023) across the seven major markets?
- What would be the forecasted patient pool (2024-2034) across the seven major markets?
- What are the key factors driving the epidemiological trend of anaplastic astrocytoma?
- What will be the growth rate of patients across the seven major markets?
Anaplastic Astrocytoma: Current Treatment Scenario, Marketed Drugs and Emerging Therapies
- What are the current marketed drugs and what are their market performance?
- What are the key pipeline drugs and how are they expected to perform in the coming years?
- How safe are the current marketed drugs and what are their efficacies?
- How safe are the late-stage pipeline drugs and what are their efficacies?
- What are the current treatment guidelines for anaplastic astrocytoma drugs across the seven major markets?
- Who are the key companies in the market and what are their market shares?
- What are the key mergers and acquisitions, licensing activities, collaborations, etc. related to the anaplastic astrocytoma market?
- What are the key regulatory events related to the anaplastic astrocytoma market?
- What is the structure of clinical trial landscape by status related to the anaplastic astrocytoma market?
- What is the structure of clinical trial landscape by phase related to the anaplastic astrocytoma market?
- What is the structure of clinical trial landscape by route of administration related to the anaplastic astrocytoma market?
This product will be updated with the latest data at the time of order. Consequently, dispatch time for this product will be 7-10 business days.
Table of Contents
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 133 |
| Published | May 2024 |
| Forecast Period | 2023 - 2034 |
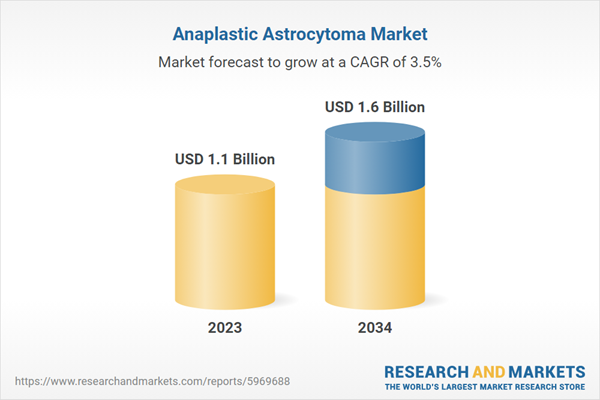
| Estimated Market Value ( USD | $ 1.1 Billion |
| Forecasted Market Value ( USD | $ 1.6 Billion |
| Compound Annual Growth Rate | 3.5% |
| Regions Covered | Global |