The neuroblastoma market has been comprehensively analyzed in this report titled "Neuroblastoma Market: Epidemiology, Industry Trends, Share, Size, Growth, Opportunity, and Forecast 2024-2034". Neuroblastoma refers to a type of cancer that develops in the immature nerve cells (neuroblasts) of the sympathetic nervous system. It usually starts in the adrenal glands, which are located above the kidneys but can also develop in other parts of the body, such as the neck, chest, abdomen, pelvis, etc. The disease generally affects infants and young children, although it can occur in adults as well. Some common symptoms are a lump in the abdomen, which may result in abdominal pain or swelling and lead to constipation or difficulty urinating. Various other indications may include bone pain, breathing problems, unexplained weight loss, fatigue, etc. In some cases, neuroblastoma may also cause fever, high blood pressure, or rapid heartbeat. The diagnosis of the ailment usually begins with a physical exam and a review of the patient's medical history. Several investigations may be used to confirm the prognosis, including imaging studies such as X-rays, ultrasounds, CT scans, MRI scans, etc., which can aid in identifying the size and location of the tumor. Additionally, numerous blood and urine tests may also be utilized to check for certain substances that are often elevated in patients with neuroblastoma. A biopsy is also performed to help confirm the diagnosis.
The increasing prevalence of genetic mutations that affect the development of nerve cells and the rising cases of chromosomal abnormalities, such as changes in the number or structure of chromosomes, are primarily driving the neuroblastoma market. In addition to this, the emerging popularity of proton therapy, since it is less damaging to the surrounding healthy tissues than traditional radiation therapy and helps treat neuroblastoma located near vital organs, is acting as another significant growth-inducing factor. Furthermore, the inflating application of gene expression profiling (GEP) to help identify the genetic signature of neuroblastoma and predict the prognosis and response to treatment is creating a positive outlook for the market. Moreover, several key players are making extensive investments in R&D activities to introduce targeted therapies that attack cancer cells while leaving healthy cells untouched, thereby improving clinical outcomes and reducing side effects. This, in turn, is also propelling the market growth. Additionally, the escalating demand for combination therapies which offer greater efficacy than single therapies alone and lower the risk of resistance developing to a single treatment is expected to drive the neuroblastoma market in the coming years.
This report provides an exhaustive analysis of the neuroblastoma market in the United States, EU5 (Germany, Spain, Italy, France, and United Kingdom) and Japan. This includes treatment practices, in-market, and pipeline drugs, share of individual therapies, market performance across the seven major markets, market performance of key companies and their drugs, etc. The report also provides the current and future patient pool across the seven major markets. According to the report the United States has the largest patient pool for neuroblastoma and also represents the largest market for its treatment. Furthermore, the current treatment practice/algorithm, market drivers, challenges, opportunities, reimbursement scenario and unmet medical needs, etc. have also been provided in the report. This report is a must-read for manufacturers, investors, business strategists, researchers, consultants, and all those who have any kind of stake or are planning to foray into the neuroblastoma market in any manner.
Time Period of the Study
- Base Year: 2023
- Historical Period: 2018-2023
- Market Forecast: 2024-2034
Countries Covered
- United States
- Germany
- France
- United Kingdom
- Italy
- Spain
- Japan
Analysis Covered Across Each Country
- Historical, current, and future epidemiology scenario
- Historical, current, and future performance of the neuroblastoma market
- Historical, current, and future performance of various therapeutic categories in the market
- Sales of various drugs across the neuroblastoma market
Competitive Landscape:
This report also provides a detailed analysis of the current neuroblastoma marketed drugs and late-stage pipeline drugs.In-Market Drugs
- Drug Overview
- Mechanism of Action
- Regulatory Status
- Clinical Trial Results
- Drug Uptake and Market Performance
Late-Stage Pipeline Drugs
- Drug Overview
- Mechanism of Action
- Regulatory Status
- Clinical Trial Results
- Drug Uptake and Market Performance
Key Questions Answered in this Report:
Market Insights
- How has the neuroblastoma market performed so far and how will it perform in the coming years?
- What are the markets shares of various therapeutic segments in 2023 and how are they expected to perform till 2034?
- What was the country-wise size of the neuroblastoma market across the seven major markets in 2023 and what will it look like in 2034?
- What is the growth rate of the neuroblastoma market across the seven major markets and what will be the expected growth over the next ten years?
- What are the key unmet needs in the market?
Epidemiology Insights
- What is the number of prevalent cases (2018-2034) of neuroblastoma across the seven major markets?
- What is the number of prevalent cases (2018-2034) of neuroblastoma by age across the seven major markets?
- What is the number of prevalent cases (2018-2034) of neuroblastoma by gender across the seven major markets?
- How many patients are diagnosed (2018-2034) with neuroblastoma across the seven major markets?
- What is the size of the neuroblastoma patient pool (2018-2023) across the seven major markets?
- What would be the forecasted patient pool (2024-2034) across the seven major markets?
- What are the key factors driving the epidemiological trend of neuroblastoma?
- What will be the growth rate of patients across the seven major markets?
Genital warts: Current Treatment Scenario, Marketed Drugs and Emerging Therapies
- What are the current marketed drugs and what are their market performance?
- What are the key pipeline drugs and how are they expected to perform in the coming years?
- How safe are the current marketed drugs and what are their efficacies?
- How safe are the late-stage pipeline drugs and what are their efficacies?
- What are the current treatment guidelines for neuroblastoma drugs across the seven major markets?
- Who are the key companies in the market and what are their market shares?
- What are the key mergers and acquisitions, licensing activities, collaborations, etc. related to the neuroblastoma market?
- What are the key regulatory events related to the neuroblastoma market?
- What is the structure of clinical trial landscape by status related to the neuroblastoma market?
- What is the structure of clinical trial landscape by phase related to the neuroblastoma market?
- What is the structure of clinical trial landscape by route of administration related to the neuroblastoma market?
This product will be updated with the latest data at the time of order. Consequently, dispatch time for this product will be 7-10 business days.
Table of Contents
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 130 |
| Published | May 2024 |
| Forecast Period | 2023 - 2034 |
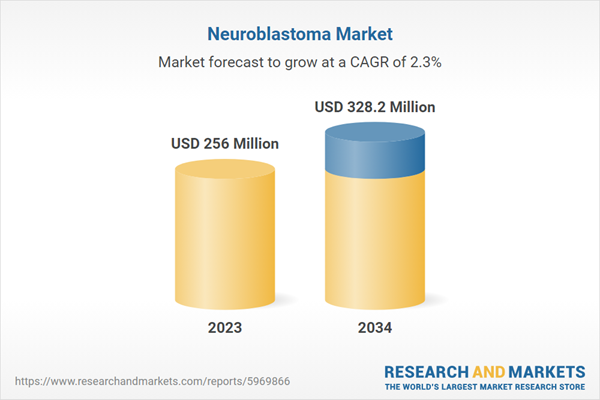
| Estimated Market Value ( USD | $ 256 Million |
| Forecasted Market Value ( USD | $ 328.2 Million |
| Compound Annual Growth Rate | 2.3% |
| Regions Covered | Global |