Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
Technological progress in plasma fractionation, purification, and the development of novel therapies is transforming the market. The outlook remains positive, with continued growth anticipated due to advancements in therapy technologies, heightened demand for specialized treatments, and supportive healthcare policies. However, market participants must navigate regulatory challenges, address supply chain constraints, and manage high production costs to leverage emerging opportunities and sustain market expansion.
Key Market Drivers
Increasing Prevalence of Blood Disorders
The growing number of individuals diagnosed with blood disorders, such as hemophilia, immune deficiencies, and other plasma-related conditions, directly increases the demand for plasma-derived therapies. As more patients require treatment for these conditions, the need for blood plasma products, such as clotting factor concentrates and immunoglobulins, intensifies.This heightened demand leads to expanded market opportunities for manufacturers and suppliers of plasma-derived products. Blood disorders often necessitate ongoing and specialized treatment regimens. For example, hemophilia patients require regular infusions of clotting factors to manage bleeding episodes, while individuals with immune deficiencies may need intravenous immunoglobulins to bolster their immune system. The chronic and long-term nature of these treatments ensures a sustained demand for plasma-derived products, driving continuous growth in the market.
The increasing prevalence of blood disorders shifts the focus of healthcare systems towards better management and treatment of these conditions. In Japan, this focus leads to the development and implementation of more comprehensive treatment protocols and guidelines. As healthcare providers and institutions prioritize these conditions, they drive the adoption and utilization of advanced plasma therapies, further expanding the market. With the rise in blood disorder prevalence, there is also an advancement in diagnostic techniques and early detection methods. Improved diagnostic capabilities lead to more accurate and earlier diagnoses, which in turn increases the number of patients identified and treated.
Early and accurate diagnosis necessitates timely access to plasma-derived therapies, contributing to the growth of the blood plasma market. The growing patient population with blood disorders stimulates research and development activities aimed at improving plasma-based treatments. Increased prevalence creates a larger patient pool for clinical trials and drives innovation in the development of new and more effective plasma-derived therapies. This focus on research and development not only enhances treatment options but also contributes to the market's growth by introducing novel products and technologies.
The rising prevalence of blood disorders encourages the establishment and expansion of specialized treatment facilities and centers dedicated to managing these conditions. These facilities, such as hemophilia treatment centers, are equipped to handle complex treatment regimens and provide access to advanced plasma therapies. The growth of such facilities supports the increased demand for plasma products and contributes to market expansion. Higher prevalence of blood disorders often leads to increased awareness and advocacy efforts. Organizations and patient advocacy groups work to raise awareness about these conditions and the importance of accessing appropriate treatments. This heightened awareness drives patient engagement and demand for plasma-derived therapies, further fueling market growth.
Advancements in Plasma-Based Therapies
Innovations in plasma-based therapies have led to the development of more effective treatments. Advances such as extended half-life clotting factors and high-potency immunoglobulins enhance therapeutic efficacy by reducing the frequency of infusions and improving clinical outcomes. These advancements not only better manage conditions such as hemophilia and immune deficiencies but also increase patient adherence to treatment regimens, driving higher demand for these advanced therapies and contributing to market growth. Technological improvements in plasma-based therapies often result in treatments that are more convenient and less invasive. For example, extended half-life products reduce the need for frequent dosing, which enhances patient compliance and satisfaction. As patients experience fewer interruptions in their daily lives and better management of their conditions, the demand for these advanced plasma therapies grows, driving market expansion.Advances in plasma-based therapies have broadened the range of conditions that can be treated. For instance, new formulations and delivery methods have expanded the use of plasma products beyond traditional indications to include rare and complex disorders. This expansion increases the overall market for plasma-derived products by tapping into new therapeutic areas and patient populations. Innovations in plasma fractionation and purification technologies have improved the efficiency and safety of plasma-derived product manufacturing. Enhanced manufacturing processes, such as advanced chromatography techniques and automated systems, increase yield, reduce production costs, and ensure higher product quality.
These improvements facilitate the scalability and reliability of plasma supply, supporting market growth by meeting the rising demand for high-quality therapies. The development of novel therapies, including gene therapies and new biologics, represents a significant advancement in the field of plasma-based treatments. These cutting-edge therapies offer potential cures or more effective management options for previously challenging conditions. The introduction of such innovations drives market growth by attracting interest from healthcare providers and patients seeking the latest and most effective treatment options.
The focus on advancing plasma-based therapies spurs significant investment in research and development (R&D). Pharmaceutical companies and research institutions invest heavily in developing new therapies, exploring innovative applications, and improving existing products. This R&D activity not only leads to the introduction of new and improved plasma-based therapies but also stimulates market growth by creating a pipeline of future products and maintaining a competitive landscape. Regulatory agencies in Japan support the advancement of plasma-based therapies by facilitating the approval of innovative products and streamlining regulatory processes. The positive regulatory environment encourages the development and commercialization of new therapies, which contributes to market growth by increasing the availability of advanced plasma products.
Rising Awareness and Diagnosis
Enhanced awareness among healthcare professionals and the general public about blood disorders leads to more frequent and accurate diagnoses. As awareness campaigns and educational programs shed light on conditions such as hemophilia, immune deficiencies, and other disorders requiring plasma-derived therapies, the rate of early and accurate detection rises. This increased detection translates into a higher number of diagnosed patients, thereby driving demand for blood plasma products as these patients require ongoing treatment.With better awareness and diagnostic capabilities, blood disorders are identified at earlier stages. Early diagnosis allows for timely intervention and management, which can prevent complications and improve patient outcomes. The availability of effective plasma-derived therapies ensures that early-diagnosed patients receive the necessary treatments, further boosting the demand for these products. Early intervention also increases the likelihood of patients adhering to long-term treatment plans, sustaining the need for plasma therapies over time.
Rising awareness often leads to the implementation of expanded screening programs for blood disorders. For example, routine screening for conditions such as hemophilia in newborns or high-risk populations ensures that more individuals are identified and treated. These screening programs increase the number of patients in need of plasma-derived treatments, contributing to market growth by broadening the patient base and enhancing the utilization of blood plasma products.
Increased awareness contributes to better education for both patients and healthcare providers regarding the benefits and availability of plasma-derived therapies. Patient education empowers individuals to seek timely medical care and adhere to prescribed treatments, while healthcare provider education ensures that medical professionals are well-informed about the latest treatment options and diagnostic practices. This dual education effort enhances the uptake of plasma therapies and drives market demand.
The rise in awareness often involves the active involvement of patient advocacy groups and support organizations. These groups play a crucial role in promoting knowledge about blood disorders and advocating for improved treatment options. Their efforts help to raise awareness, support research, and influence policy changes, all of which contribute to increased demand for plasma-derived therapies and drive market growth. As awareness of blood disorders grows, there is often an increase in funding and resources allocated to research, treatment, and support services.
Governments, non-profit organizations, and private entities may invest more in research and development of new plasma-based therapies, diagnostic tools, and patient support programs. This influx of funding supports innovation and enhances the availability and effectiveness of plasma products, driving market expansion. Growing awareness leads to the development of comprehensive disease management programs that focus on optimizing patient care. These programs often include regular monitoring, personalized treatment plans, and coordination between healthcare providers. Effective disease management programs improve patient outcomes and encourage the consistent use of plasma-derived therapies, thereby increasing their demand and contributing to market growth.
Key Market Challenges
Regulatory and Compliance Issues
The blood plasma market in Japan is subject to stringent regulatory requirements and compliance standards. The Japanese government and regulatory bodies impose rigorous controls on the collection, processing, and distribution of blood plasma products to ensure safety and efficacy. Navigating these complex regulations can be challenging for companies, potentially leading to delays in product approvals and increased operational costs. Additionally, compliance with international regulations and standards for plasma products can add further complexity and cost. These regulatory hurdles can impede market entry for new players and slow down the introduction of innovative plasma therapies, restricting overall market growth.Supply Chain Constraints
The blood plasma supply chain is highly intricate and can be vulnerable to disruptions. Challenges such as limited availability of high-quality plasma donors, logistical issues in plasma collection and transportation, and the need for sophisticated processing and storage facilities can impact the reliability and efficiency of the supply chain. In Japan, where the demand for plasma products is high, ensuring a consistent and adequate supply of plasma is critical. Supply chain constraints can lead to shortages, affect product availability, and increase costs, which can hinder market growth and limit the ability of companies to meet patient needs effectively.High Production Costs
The production of blood plasma-derived products involves complex and costly processes, including plasma collection, fractionation, purification, and formulation. The high costs associated with these processes can pose significant challenges for manufacturers. In Japan, where advanced technologies and high-quality standards are required, production costs can be particularly elevated. These costs are often passed on to consumers and healthcare providers, which can limit access to plasma therapies and impact market growth. Furthermore, the substantial investment required for research and development of new products adds to the financial burden, potentially deterring investment and slowing the introduction of innovative therapies.Key Market Trends
Advancements in Treatment Technologies
Significant progress in blood plasma treatment technologies is a major driver of market growth. Innovations such as recombinant clotting factors, extended half-life products, and advanced fractionation methods are enhancing the efficacy and convenience of plasma-derived therapies. These technological advancements improve patient outcomes and reduce the frequency of treatments required, thereby expanding the market for blood plasma products. Additionally, the development of novel therapies, including gene therapies and biologics, promises to revolutionize treatment approaches and cater to previously unmet medical needs. The ongoing evolution in treatment technologies is expected to drive demand and foster market expansion.Increasing Demand for Personalized Medicine
The growing emphasis on personalized medicine is another key trend influencing the blood plasma market. Personalized medicine aims to tailor treatments to individual patient profiles based on genetic, environmental, and lifestyle factors. In the context of blood plasma, this trend translates into the development of more targeted and individualized therapies. Advances in genomic and molecular research are enabling the creation of customized plasma-derived products that address specific patient needs more effectively. This shift towards personalized approaches is driving the demand for advanced plasma therapies and is expected to significantly contribute to market growth as healthcare providers seek to optimize treatment outcomes for their patients.Strategic Market Expansions
Expansion strategies by leading plasma product manufacturers and distributors are playing a crucial role in driving market growth. Companies are increasingly focusing on expanding their market presence through strategic partnerships, acquisitions, and regional expansions. This includes establishing new production facilities, enhancing distribution networks, and entering emerging markets. In Japan, this trend is reflected in the increased investment in domestic manufacturing capabilities and the development of more efficient supply chains. By broadening their market reach and improving accessibility to plasma products, companies are positioning themselves to capitalize on growing demand and emerging opportunities within the Japanese healthcare landscape.Segmental Insights
Type Insights
Based on the category of Type, the Immunoglobulins segment emerged as the dominant in the market for Japan Blood Plasma in 2024. Immunoglobulins, particularly intravenous immunoglobulins (IVIG), are critical for treating a range of conditions, including primary immunodeficiency disorders, autoimmune diseases, and chronic inflammatory conditions. The prevalence of these conditions in Japan drives substantial demand for immunoglobulin products.This high clinical demand positions immunoglobulins as a leading segment in the blood plasma market. Applications: Immunoglobulins are utilized in various advanced therapeutic applications. They play a pivotal role in treating complex medical conditions such as Guillain-Barré syndrome, chronic inflammatory demyelinating polyneuropathy, and Kawasaki disease. The effectiveness of immunoglobulins in these high-impact therapeutic areas underpins their significant market share.
The immunoglobulin segment benefits from ongoing advancements in manufacturing technologies and processes. Innovations such as improved fractionation techniques and more efficient purification methods enhance the yield and quality of immunoglobulins. These advancements contribute to the segment's dominance by ensuring a consistent supply of high-quality products. The Japanese regulatory framework provides robust support for the development and commercialization of immunoglobulin products.
Additionally, favorable reimbursement policies for immunoglobulin therapies ensure broad patient access and healthcare provider support, further bolstering the segment’s market position. Immunoglobulins are well-integrated into clinical guidelines and treatment protocols in Japan. This established role in standard medical practice ensures their continued use and drives sustained demand. The clear clinical guidelines for the use of immunoglobulins reinforce their importance in the treatment landscape.
Significant investment in research and development for immunoglobulin therapies has led to the introduction of new and enhanced products. These investments drive innovation and contribute to the growth of the immunoglobulin segment, reinforcing its dominance in the blood plasma market. The proven efficacy of immunoglobulins in improving patient outcomes further solidifies their market position. Positive clinical outcomes and high patient satisfaction with immunoglobulin therapies drive continued preference and use in healthcare settings, enhancing the segment’s prominence. These factors are expected to drive the growth of this segment.
Application Insights
The Hemophilia segment is projected to experience rapid growth during the forecast period. Hemophilia, a genetic bleeding disorder characterized by the deficiency of clotting factors, presents a significant clinical challenge in Japan. The condition requires regular and specific treatment with clotting factor concentrates derived from blood plasma. The high prevalence of hemophilia and the necessity for ongoing management drives substantial demand for plasma-derived products within this application segment.Recent advancements in the treatment of hemophilia, including the development of more effective and longer-lasting clotting factor products, have significantly impacted the market. Innovations such as recombinant factor therapies and extended half-life products have improved treatment outcomes and reduced the frequency of infusions, leading to increased utilization of these products and reinforcing the dominance of the hemophilia segment. Japan has a well-developed healthcare infrastructure dedicated to the management of hemophilia. This includes specialized hemophilia treatment centers (HTCs) and multidisciplinary teams focused on comprehensive care for hemophilia patients. The presence of such specialized facilities enhances the diagnosis, treatment, and management of hemophilia, driving the demand for blood plasma-derived clotting factors.
The Japanese healthcare system provides strong support for the treatment of hemophilia, including comprehensive insurance coverage for clotting factor therapies. Government initiatives and favorable reimbursement policies ensure that patients have access to necessary treatments, which supports the growth of the hemophilia segment in the blood plasma market. Increased awareness among patients, caregivers, and healthcare professionals about the importance of effective hemophilia management contributes to the segment's dominance. Educational efforts and advocacy initiatives emphasize the benefits of advanced clotting factor therapies, leading to higher adoption rates and sustained demand. These factors collectively contribute to the growth of this segment.
Regional Insights
Kanto emerged as the dominant in the Japan Blood Plasma market in 2024, holding the largest market share in terms of value. The Kanto region, encompassing major metropolitan areas such as Tokyo and Yokohama, boasts a well-established and extensive healthcare infrastructure. This includes a high concentration of hospitals, specialized clinics, and research institutions that are crucial for the collection, processing, and utilization of blood plasma.The presence of these facilities facilitates efficient plasma procurement and distribution, meeting the region's substantial demand. As Japan's economic powerhouse, the Kanto region benefits from significant investment in healthcare and biotechnology sectors. This economic strength allows for advanced research and development in blood plasma-related therapies and technologies. Companies in this region often lead innovations in plasma fractionation and related treatments, which further propels their market dominance.
The Kanto region has the largest population in Japan, contributing to a high incidence of conditions requiring blood plasma-derived therapies, such as hemophilia and immunodeficiency disorders. This substantial patient base drives a higher demand for plasma products, ensuring that the Kanto region remains a focal point for plasma supply and utilization.
The Kanto region benefits from a robust regulatory framework and stringent quality standards, which are essential for the production and distribution of blood plasma products. The region's adherence to these high standards ensures the safety and efficacy of plasma-derived therapies, which is critical for maintaining trust and sustaining market growth. The Kanto region's strategic location as a transportation hub facilitates the efficient movement of blood plasma products. Its well-developed infrastructure, including ports and transportation networks, supports the logistics and supply chain necessary for the timely delivery of plasma products across Japan and internationally.
Key Market Players
- CSL
- Grifols, S.A.
- Octapharma AG
- Biotest AG
- Kedrion S.p.A
Report Scope:
In this report, the Japan Blood Plasma Market has been segmented into the following categories, in addition to the industry trends which have also been detailed below:Japan Blood Plasma Market, By Type:
- Albumin
- Factor VIII
- Factor IX
- Immunoglobulin
- Hyperimmune Globulin
- Others
Japan Blood Plasma Market, By Application:
- Hemophilia
- Hypogammaglobulinemia
- Immunodeficiency Diseases
- Von Willebrand’s Disease
- Others
Japan Blood Plasma Market, By End User:
- Hospitals
- Clinics
- Others
Japan Blood Plasma Market, By Region:
- Hokkaido
- Tohoku
- Kanto
- Chubu
- Kansai
- Chugoku
- Shikoku
- Kyushu
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Japan Blood Plasma Market.Available Customizations:
Japan Blood Plasma market report with the given market data, the publisher offers customizations according to a company's specific needs. The following customization options are available for the report:Company Information
- Detailed analysis and profiling of additional market players (up to five).
This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
- CSL
- Grifols, S.A.
- Octapharma AG
- Biotest AG
- Kedrion S.p.A
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 82 |
| Published | September 2024 |
| Forecast Period | 2024 - 2030 |
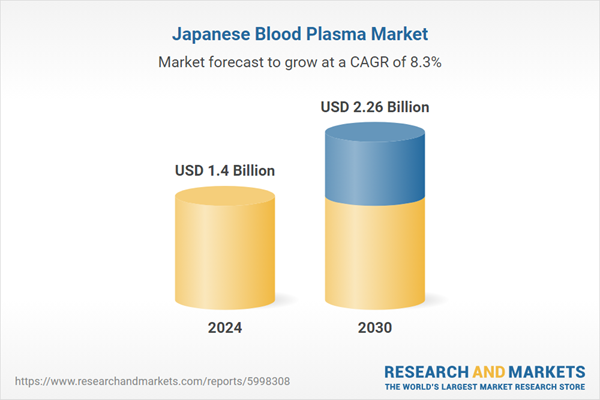
| Estimated Market Value ( USD | $ 1.4 Billion |
| Forecasted Market Value ( USD | $ 2.26 Billion |
| Compound Annual Growth Rate | 8.2% |
| Regions Covered | Japan |
| No. of Companies Mentioned | 5 |