Atrial Fibrillation Devices - Key Trends and Drivers
Atrial fibrillation (AF) is the most common type of cardiac arrhythmia, characterized by rapid and irregular beating of the atrial chambers of the heart. This condition significantly increases the risk of stroke, heart failure, and other cardiovascular complications. To manage and treat AF, a variety of medical devices are used, including catheter ablation devices, pacemakers, implantable cardioverter defibrillators (ICDs), and left atrial appendage (LAA) closure devices. Catheter ablation, in particular, has emerged as a highly effective treatment for AF, involving the delivery of energy through a catheter to destroy the heart tissue causing the irregular rhythm. Pacemakers and ICDs are used to regulate heart rhythms and prevent sudden cardiac arrest, while LAA closure devices help reduce the risk of stroke by sealing off the area of the heart where blood clots commonly form in AF patients.Recent advancements in atrial fibrillation devices have significantly improved their efficacy and safety. Innovations in catheter ablation technology, such as the development of cryoablation and laser ablation, offer more precise and less invasive options for patients. The introduction of contact force-sensing catheters has enhanced the precision of ablation procedures, ensuring more effective tissue destruction while minimizing damage to surrounding areas. Moreover, advancements in imaging and mapping technologies have enabled better visualization of the heart's anatomy and electrical activity, leading to more accurate and successful ablation treatments. The miniaturization and enhanced battery life of pacemakers and ICDs have made these devices more patient-friendly, reducing the need for frequent replacements and allowing for less invasive implantation procedures. Additionally, the design improvements in LAA closure devices have resulted in higher success rates and lower complication rates.
The growth in the atrial fibrillation devices market is driven by several factors. Advances in medical technology, such as the development of next-generation ablation catheters and improved imaging systems, are significantly enhancing the outcomes of AF treatments and driving the adoption of these devices. The increasing prevalence of atrial fibrillation, particularly among the aging population, is expanding the patient base requiring treatment. Furthermore, growing awareness about the risks associated with untreated AF and the availability of advanced treatment options are encouraging more patients to seek medical intervention. Rise in healthcare expenditure and improved access to healthcare services in developing regions are also contributing to market growth. Additionally, strategic collaborations and partnerships among key players in the medical device industry are fostering innovation and expanding the availability of advanced AF devices. The integration of artificial intelligence (AI) and machine learning (ML) in the development and deployment of these devices is further propelling the market, offering enhanced diagnostic and therapeutic capabilities. These factors collectively ensure the robust growth and continued evolution of the atrial fibrillation devices market.
Report Scope
The report analyzes the Atrial Fibrillation Devices market, presented in terms of market value. The analysis covers the key segments and geographic regions outlined below.- Segments: Type (EP Ablation Catheters, EP Diagnostic Catheters, EP Mapping & Recording Systems, Cardiac Monitors, Access Devices, LAA Closure, Other Types); Application (Diagnostics, EP Ablation, Surgical); End-Use (Hospitals, Electrophysiology Labs, Ambulatory Surgery Centers).
- Geographic Regions/Countries: World; United States; Canada; Japan; China; Europe (France; Germany; Italy; United Kingdom; Spain; Russia; and Rest of Europe); Asia-Pacific (Australia; India; South Korea; and Rest of Asia-Pacific); Latin America (Argentina; Brazil; Mexico; and Rest of Latin America); Middle East (Iran; Israel; Saudi Arabia; United Arab Emirates; and Rest of Middle East); and Africa.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the EP Ablation Catheters segment, which is expected to reach US$4.8 Billion by 2030 with a CAGR of a 14.5%. The EP Diagnostic Catheters segment is also set to grow at 11.3% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $2.1 Billion in 2024, and China, forecasted to grow at an impressive 16.9% CAGR to reach $3.8 Billion by 2030. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global Atrial Fibrillation Devices Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global Atrial Fibrillation Devices Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global Atrial Fibrillation Devices Market expected to evolve by 2030?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2030?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2024 to 2030.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as Acceleron Pharma, Inc., Amgen, Inc., Bayer AG, Biocon Ltd., bluebird bio, Inc. and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the 34 companies featured in this Atrial Fibrillation Devices market report include:
- Abbott Laboratories, Inc.
- AtriCure, Inc.
- Biotronik SE & Co. KG
- Boston Scientific Corporation
- CardioFocus, Inc.
- Johnson & Johnson
- Koninklijke Philips NV
- Medtronic PLC
- MicroPort Scientific Corporation
- OSYPKA AG
This edition integrates the latest global trade and economic shifts into comprehensive market analysis. Key updates include:
- Tariff and Trade Impact: Insights into global tariff negotiations across 180+ countries, with analysis of supply chain turbulence, sourcing disruptions, and geographic realignment. Special focus on 2025 as a pivotal year for trade tensions, including updated perspectives on the Trump-era tariffs.
- Adjusted Forecasts and Analytics: Revised global and regional market forecasts through 2030, incorporating tariff effects, economic uncertainty, and structural changes in globalization. Includes historical analysis from 2015 to 2023.
- Strategic Market Dynamics: Evaluation of revised market prospects, regional outlooks, and key economic indicators such as population and urbanization trends.
- Innovation & Technology Trends: Latest developments in product and process innovation, emerging technologies, and key industry drivers shaping the competitive landscape.
- Competitive Intelligence: Updated global market share estimates for 2025, competitive positioning of major players (Strong/Active/Niche/Trivial), and refined focus on leading global brands and core players.
- Expert Insight & Commentary: Strategic analysis from economists, trade experts, and domain specialists to contextualize market shifts and identify emerging opportunities.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories, Inc.
- AtriCure, Inc.
- Biotronik SE & Co. KG
- Boston Scientific Corporation
- CardioFocus, Inc.
- Johnson & Johnson
- Koninklijke Philips NV
- Medtronic PLC
- MicroPort Scientific Corporation
- OSYPKA AG
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 379 |
| Published | February 2026 |
| Forecast Period | 2024 - 2030 |
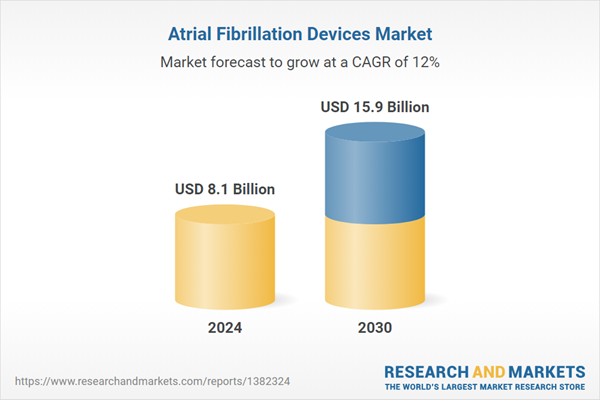
| Estimated Market Value ( USD | $ 8.1 Billion |
| Forecasted Market Value ( USD | $ 15.9 Billion |
| Compound Annual Growth Rate | 12.0% |
| Regions Covered | Global |