Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
In North America, the market for dissolution testing services is witnessing steady growth due to the increasing complexity of drug formulations, stringent regulatory requirements, and a growing emphasis on ensuring bioequivalence and therapeutic efficacy. The pharmaceutical dissolution testing services encompass a range of offerings, including method development, validation, and routine testing to assess the performance of solid oral dosage forms. The market is characterized by the presence of specialized laboratories and contract research organizations (CROs) equipped with advanced instrumentation and expertise in dissolution testing methodologies.
Key factors driving the market include the rising demand for generic drugs, the need for comprehensive quality control measures, and the increasing outsourcing of pharmaceutical testing services by drug manufacturers. Regulatory compliance remains a central focus, with pharmaceutical companies relying on dissolution testing services to meet the requirements set by health authorities such as the U.S. Food and Drug Administration (FDA). Advancements in technology, such as automation and sophisticated analytical techniques, contribute to the efficiency and accuracy of dissolution testing services. As pharmaceutical companies strive to optimize their drug development processes and ensure timely regulatory approvals, the North America Pharmaceutical Dissolution Testing Services Market is poised for continued expansion, offering vital support to the region's pharmaceutical research and manufacturing landscape.
Key Market Drivers
Increasing Complexity of Drug Formulations
The North America Pharmaceutical Dissolution Testing Services Market is experiencing a significant upswing propelled by the increasing complexity of drug formulations. As pharmaceutical companies strive for innovation to develop more effective and targeted therapies, the landscape of drug formulations has evolved to encompass intricate structures and novel delivery systems. The demand for dissolution testing services has surged in response to this complexity, as these services play a pivotal role in evaluating how active pharmaceutical ingredients (APIs) release over time from diverse and sophisticated formulations.Modern drug formulations include extended-release formulations, multi-particulate systems, and complex combinations of APIs and excipients. The intricate nature of these formulations necessitates specialized testing methodologies and expertise to ensure accurate assessments of their dissolution behavior. This trend is particularly prominent in North America, where pharmaceutical companies are navigating the challenges posed by intricate formulations while striving to meet rigorous regulatory standards. Dissolution testing services, equipped with advanced instrumentation and skilled professionals, are essential in providing precise insights into the dissolution profiles of these complex drug formulations.
The surge in demand for dissolution testing services aligns with the pharmaceutical industry's commitment to maintaining product quality, safety, and efficacy. These services not only assist in the early stages of drug development but also play a crucial role in post-market quality control and batch-to-batch consistency. As the complexity of drug formulations continues to increase, the North America Pharmaceutical Dissolution Testing Services Market is well-positioned to be a cornerstone in ensuring that pharmaceutical products meet the evolving standards of regulatory agencies and deliver the desired therapeutic outcomes. The adaptability of testing services to handle diverse formulations underscores their importance in a pharmaceutical landscape where innovation and complexity are driving forces in shaping the future of drug development and patient care.
Growing Emphasis on Bioequivalence Studies
The North America Pharmaceutical Dissolution Testing Services Market has experienced a significant boost in recent years, primarily attributed to the growing emphasis on bioequivalence studies. Bioequivalence studies play a crucial role in the pharmaceutical industry, ensuring that generic drugs are equivalent to their brand-name counterparts in terms of safety, efficacy, and quality.As regulatory agencies continue to prioritize and mandate stringent bioequivalence requirements, pharmaceutical companies are increasingly turning to dissolution testing services to meet these standards. Dissolution testing is a critical component of bioequivalence studies, providing insights into how a drug dissolves and releases its active ingredients in the body. The demand for accurate and reliable dissolution testing services has surged, driving the growth of specialized service providers in North America.
The pharmaceutical industry's recognition of the pivotal role that dissolution testing plays in drug development and regulatory approval has led to a surge in outsourcing these services to specialized providers. These providers offer state-of-the-art facilities, advanced technologies, and expertise in conducting dissolution testing in compliance with regulatory guidelines. This trend is particularly pronounced in North America, where pharmaceutical companies are keen on leveraging external expertise to navigate the complex landscape of bioequivalence studies. The outsourcing of dissolution testing services allows pharmaceutical companies to focus on their core competencies, accelerate drug development timelines, and ensure compliance with evolving regulatory standards.
The increased adoption of innovative formulations, such as extended-release and controlled-release formulations, has further propelled the demand for dissolution testing services. These formulations require sophisticated testing methodologies to assess their release profiles accurately. As pharmaceutical companies strive to bring novel and advanced drug formulations to market, the reliance on specialized dissolution testing services is expected to continue its upward trajectory.
Advancements in Technology and Automation
The North America Pharmaceutical Dissolution Testing Services Market is experiencing a notable surge, propelled by significant advancements in technology and automation. In recent years, the pharmaceutical industry has witnessed a paradigm shift towards adopting cutting-edge technologies to enhance the efficiency, accuracy, and reliability of dissolution testing processes. Automation, in particular, has emerged as a transformative force, streamlining workflows and reducing human error in pharmaceutical dissolution testing. Automated dissolution testing systems not only increase the precision of results but also contribute to higher throughput, enabling pharmaceutical companies to handle larger volumes of samples more efficiently.Technological innovations have introduced novel techniques and instruments that are revolutionizing dissolution testing methodologies. For instance, the integration of artificial intelligence (AI) and machine learning (ML) algorithms into dissolution testing processes has enabled real-time data analysis, predictive modeling, and adaptive control. This not only expedites the testing phase but also enhances the overall quality of the results. The ability of these advanced technologies to optimize dissolution testing parameters and identify patterns in dissolution profiles has become instrumental in ensuring the accuracy and reliability of drug release assessments.
The incorporation of robotics in sample preparation, sample loading, and data collection has significantly automated the entire dissolution testing workflow. Robotic systems contribute to precision and repeatability, reducing variability in test results. This level of automation is particularly attractive to pharmaceutical companies aiming to improve operational efficiency, minimize resource requirements, and meet stringent regulatory standards. The integration of robotic platforms has not only accelerated testing processes but has also contributed to the standardization of dissolution testing methodologies across the industry.
The North America Pharmaceutical Dissolution Testing Services Market is witnessing a growing trend of pharmaceutical companies outsourcing their dissolution testing needs to specialized service providers equipped with state-of-the-art automated technologies. These providers offer a combination of expertise and advanced instrumentation, ensuring that pharmaceutical companies can access the latest innovations in dissolution testing without making substantial capital investments.
Key Market Challenges
Regulatory Complexity and Evolving Standards
The North America Pharmaceutical Dissolution Testing Services Market faces a significant hurdle in the form of regulatory complexity and evolving standards. The pharmaceutical industry operates in a highly regulated environment, with stringent guidelines set by regulatory bodies such as the U.S. Food and Drug Administration (FDA). These regulations play a crucial role in ensuring the safety, efficacy, and quality of pharmaceutical products. However, the ever-evolving nature of these standards poses a challenge for both pharmaceutical companies and dissolution testing service providers.The regulatory landscape is dynamic, with updates and revisions to guidelines occurring regularly. This constant state of flux requires stakeholders to stay vigilant and adapt swiftly to changes. For dissolution testing services, this means implementing new methodologies, adjusting testing protocols, and ensuring compliance with the latest regulatory requirements. The complexity of these standards can be a significant barrier, particularly for smaller testing laboratories or companies without dedicated regulatory affairs teams.
The challenge intensifies when considering the global nature of the pharmaceutical industry. Harmonizing standards across different regions and ensuring compliance with international regulations further adds to the intricacy of the regulatory landscape. Testing service providers in North America must contend not only with local regulations but also navigate the nuances of global regulatory frameworks.
As regulatory bodies refine their expectations for data integrity, documentation, and reporting, the burden on dissolution testing service providers increases. Adhering to these evolving standards demands a constant commitment to quality control, validation of testing methods, and meticulous documentation of processes.
High Initial Investment and Operational Costs
The North America Pharmaceutical Dissolution Testing Services Market confronts a substantial obstacle in the form of high initial investment and operational costs. The adoption of cutting-edge technologies and advanced automation systems for dissolution testing demands a significant upfront financial commitment from both pharmaceutical companies and testing service providers. The sophisticated equipment required for precise dissolution testing comes with a considerable price tag, comprising instrumentation such as automated dissolution testers, high-performance liquid chromatography (HPLC) systems, and associated software.This high initial investment acts as a deterrent, particularly for smaller pharmaceutical companies and testing laboratories with limited financial resources. The need to allocate substantial capital for state-of-the-art equipment can be a barrier to entry or expansion in the market. The complexity of the technology necessitates ongoing investments in calibration, maintenance, and upgrades to ensure optimal performance and adherence to evolving industry standards.
Operational costs add another layer to the financial challenge. Maintaining and operating advanced dissolution testing systems requires skilled personnel, ongoing training programs, and adherence to stringent quality control measures. The costs associated with personnel training, equipment maintenance, and compliance with regulatory requirements contribute to the overall operational expenses, placing a strain on the financial resources of testing service providers.
Key Market Trends
Rising Demand for Generic Drugs
The North America Pharmaceutical Dissolution Testing Services Market is experiencing a notable upswing, driven by the rising demand for generic drugs in the pharmaceutical industry. The increasing prevalence of chronic diseases and the need for cost-effective healthcare solutions have spurred the demand for generic alternatives, creating a substantial market for pharmaceutical dissolution testing services. Generic drugs must undergo rigorous bioequivalence studies to demonstrate their therapeutic equivalence to brand-name counterparts, ensuring that they meet stringent regulatory standards for safety, efficacy, and quality. Dissolution testing plays a pivotal role in these studies, providing critical insights into the release profiles of active ingredients in generic formulations.As the demand for generic drugs continues to grow, pharmaceutical companies are increasingly relying on specialized dissolution testing services to expedite the regulatory approval process. Generic drug manufacturers seek to demonstrate that their products exhibit the same dissolution characteristics as the reference listed drugs, and outsourcing dissolution testing to specialized service providers has become a strategic approach. This surge in demand for dissolution testing services is further fueled by the need for thorough and reliable assessments, as generic drug formulations often involve different excipients and manufacturing processes that must be carefully evaluated.
The competitive landscape of the pharmaceutical industry, coupled with the expiration of patents for many brand-name drugs, has led to an influx of generic alternatives. As a result, there is a growing need for efficient and high-throughput dissolution testing services to accommodate the increased volume of generic drug submissions. Specialized service providers in North America offer expertise in navigating the complexities of generic drug development, ensuring that companies can meet regulatory requirements and bring their generic products to market expeditiously.
Integration of Automation and Advanced Analytical Techniques
The North America Pharmaceutical Dissolution Testing Services Market is experiencing a substantial boost, driven by the integration of automation and advanced analytical techniques in dissolution testing processes. Automation, combined with sophisticated analytical tools, has ushered in a new era of efficiency, precision, and reliability in pharmaceutical dissolution testing. The integration of robotic systems in sample preparation, dissolution apparatus handling, and data collection has streamlined workflows, significantly reducing human error and increasing the throughput of testing processes. This level of automation not only expedites testing timelines but also ensures consistency and reproducibility of results, critical factors in meeting regulatory requirements.The incorporation of advanced analytical techniques, such as high-performance liquid chromatography (HPLC) and mass spectrometry (MS), has further enhanced the accuracy and sensitivity of dissolution testing. These techniques allow for the quantification of drug release profiles with unprecedented precision, enabling pharmaceutical companies to obtain detailed information about the dissolution behavior of their formulations. The real-time data generated by these advanced analytical tools provides valuable insights into the performance of drug products, facilitating better decision-making in the drug development process.
The integration of automation extends beyond robotic systems to include artificial intelligence (AI) and machine learning (ML) algorithms. These technologies play a crucial role in data analysis, pattern recognition, and predictive modeling, enabling the identification of subtle dissolution trends and potential issues. The ability of AI and ML to adapt and optimize dissolution testing parameters based on real-time data contributes to a more efficient and cost-effective drug development process.
Segmental Insights
Service Method Insights
Based on the method, the in-vitro method segment emerged as the dominant player in the North America market for Pharmaceutical Dissolution Testing Services in 2023. In vitro dissolution testing is a well-established and widely accepted method for assessing the performance of pharmaceutical formulations. Regulatory authorities, such as the U.S. Food and Drug Administration (FDA), often require in vitro dissolution data as part of the drug approval process.Meeting regulatory standards is a critical factor in the pharmaceutical industry, and in vitro testing provides a standardized and reproducible approach for demonstrating the release characteristics of drug formulations. In vitro testing is generally more cost-effective and efficient compared to in vivo studies, which involve testing within a living organism. In vivo studies are often more time-consuming, resource-intensive, and ethically challenging. In contrast, in vitro dissolution testing allows for quicker and more cost-efficient assessments of drug release profiles, making it a preferred method for routine quality control and formulation development.
Dosage Form Insights
Based on the Dosage Form, tablet segment emerged as the dominant player in the North America market for Pharmaceutical Dissolution Testing Services in 2023. Tablets are one of the most widely manufactured and consumed dosage forms in the pharmaceutical industry. Their popularity is attributed to factors such as ease of manufacturing, convenience of administration, stability, and patient preference. The extensive production of tablet formulations across a diverse range of therapeutic categories drives the demand for dissolution testing services. Regulatory authorities, including the U.S. Food and Drug Administration (FDA), often require dissolution testing data for tablets as part of the drug approval process.Dissolution profiles for tablets are essential to assess the release characteristics of active pharmaceutical ingredients (APIs) from the formulation, ensuring bioavailability and therapeutic efficacy. Compliance with regulatory standards contributes to the high demand for dissolution testing services, particularly for tablets. Tablets come in various formulations, including immediate-release, modified-release, and extended-release formulations. Each type requires specific dissolution testing to assess its performance. The versatility and adaptability of dissolution testing services to cater to different tablet formulations contribute significantly to their prevalence in the market.
Country Insights
The United States has one of the largest and most diverse pharmaceutical industries globally. The presence of numerous pharmaceutical companies, ranging from large multinational corporations to small and medium-sized enterprises, contributes to a substantial demand for dissolution testing services. The extensive drug development and manufacturing activities in the U.S.generate a consistent need for dissolution testing to ensure the quality, safety, and efficacy of pharmaceutical products. The U.S. regulatory environment, particularly governed by the U.S. Food and Drug Administration (FDA), is known for its stringent standards and regulatory oversight. Pharmaceutical companies operating in the United States are required to conduct comprehensive dissolution testing to meet these rigorous regulatory standards. This emphasis on regulatory compliance significantly drives the demand for dissolution testing services as an integral part of the drug development and approval processes.
Key Market Players
- Intertek Group Plc
- Avivia BV
- Almac Group
- Agilent Technologies, Inc.
- Catalent, Inc.
- Thermofisher Scientific Inc
- Charles River Laboratories
- Cambrex Corporation
- Boston Analytical Sciences
- Pace Analytical Life Sciences
Report Scope:
In this report, the North America Pharmaceutical Dissolution Testing Services Market has been segmented into the following categories, in addition to the industry trends which have also been detailed below:North America Pharmaceutical Dissolution Testing Services Market, By Services Method:
- In vitro
- In vivo
North America Pharmaceutical Dissolution Testing Services Market, By Dosage Form:
- Capsule
- Tablets
- Others
North America Pharmaceutical Dissolution Testing Services Market, By Dissolution Apparatus:
- Basket
- Paddle
- Others
North America Pharmaceutical Dissolution Testing Services Market, By Country:
- United States
- Canada
- Mexico
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the North America Pharmaceutical Dissolution Testing Services Market.Available Customizations:
With the given market data, the publisher offers customizations according to a company's specific needs. The following customization options are available for the report.Company Information
- Detailed analysis and profiling of additional market players (up to five).
This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
- Intertek Group Plc
- Avivia BV
- Almac Group
- Agilent Technologies, Inc
- Catalent, Inc
- Thermofisher Scientific Inc
- Charles River Laboratories
- Cambrex Corporation
- Boston Analytical Sciences
- Pace Analytical Life Sciences
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 131 |
| Published | December 2024 |
| Forecast Period | 2023 - 2029 |
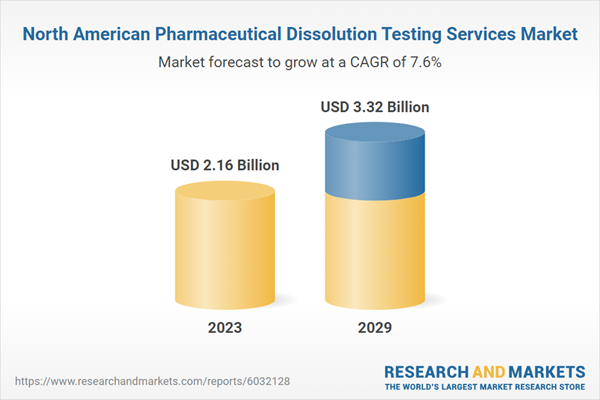
| Estimated Market Value ( USD | $ 2.16 Billion |
| Forecasted Market Value ( USD | $ 3.32 Billion |
| Compound Annual Growth Rate | 7.6% |
| Regions Covered | North America |
| No. of Companies Mentioned | 10 |